Richard Deth, Ph.D., is a neuropharmacologist and professor of pharmacology at Northeastern University in Boston, Massachusetts. Deth has published scientific studies on the role of D4 dopamine receptors in psychiatric disorders, as well as the book, Molecular Origins of Human Attention: The Dopamine-Folate Connection. His current research is directed toward understanding the roles of redox and methylation in autism, and he has served on the scientific advisory boards of the National Autism Association, the Autism Research Institute and Generation RescueFocus: You say you view life through redox glasses—not the proverbial rose-colored glasses—and that this perspective has changed how you see life and health. Can you tell us about that?
RD: In the simplest terms, maintaining life can be viewed as the ability to resist oxidation. Oxygen is essential to life, but oxygen is like fire. It can be very damaging and needs to be controlled by antioxidants, known as “reducing” molecules. Balancing reduction and oxidation—or redox–is the fundamental challenge of life. What’s great about that word, redox, is that it shows that they are profoundly linked and that we need both. Once you understand this relationship, it leads to all kinds of new insights.
Focus: What are some of those insights?
RD: The first is that, from the very moment of conception, life can be sparked by the unique redox environment created when a sperm fertilises an egg. The sperm is extremely rich in proteins containing the mineral selenium, which is a potent reducing agent for glutathione, the most important antioxidant molecule in cells. The egg, on the other hand, is very rich in glutathione. Bring these two potent antioxidant strategies together, and you create an exceptionally reduced cell that can initiate life and promote development using the power of redox. That reducing power provides a metabolic spark as new life begins its journey, allowing the rapidly dividing cells to safely maintain a high rate of oxidation. The same metabolic challenge continues as the embryo develops. The entire nervous system and the shaping of gene activity are profoundly influenced by this redox balance as well. Aging is essentially a process of gradual oxidation, and our health as we age depends on successfully quenching that oxidation. Finally, innumerable diseases are linked to high levels of oxidation and low levels of glutathione—from schizophrenia to major depression, autism, chronic fatigue syndrome, fibromyalgia, and most chronic autoimmune and chronic inflammatory diseases.
Both gluten (found in grains such as wheat) and casein (milk protein) can inhibit the uptake of cysteine, which the body needs to make glutathione.
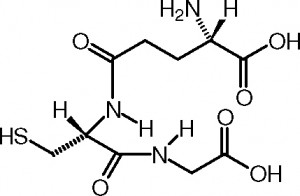 Focus: So let’s start with glutathione. How do we make this critical antioxidant, glutathione?
Focus: So let’s start with glutathione. How do we make this critical antioxidant, glutathione?
RD: The short answer is a single word: cysteine. You can get cysteine from the diet, in meat, eggs, garlic, onions, red pepper, broccoli and other foods. Cells in the gut lining, aided by transporter molecules, will bring it into the body. However, and this is a very important point, both gluten (found in grains such as wheat) and casein (milk protein) can inhibit the uptake of cysteine. Why is it that so many children with autism, or adults with autoimmune disorders, do better when they eliminate wheat and milk from their diet? I personally think it’s due to a redox mechanism.
Focus: How do two of our most popular foods inhibit uptake of such an important protein?
RD: Both casein and gluten are broken down into certain peptides that are relatively stable. The protein casein is broken into casomorphins. The “morphins” are so named because, like morphine, they act on the opiate receptors. The most famous one, beta casomorphin 7 (BCM7), has seven amino acids. Our recent research shows that BCM7 first stimulates the uptake of cysteine, but then inhibits it. However, the human BCM7 is markedly different than bovine BCM7 from the cow. It turns out that the BCM7 from a cow inhibits cysteine at least twice as much as the BCM7 from a human mother. The implications for health are profound if you start thinking about formula feeding and all the dairy products from cows in our diet. Put your redox glasses on. Breastfeeding is clearly regulating the redox system of newborns. A diet high in dairy from cows can promote a decrease in our antioxidant capacity, our ability to make enough glutathione.
Focus: What about sheep’s or goat’s milk?
RD: Our lab has looked at the peptide from sheep’s milk, and it behaves more like human milk. We haven’t studied goat’s milk yet. Similarly, the protein in gluten is known as gliadin, and it also creates a seven amino acid peptide, like BCM7. We already know that gliadin can trigger coeliac disease, and can also lead to gluten intolerance and sensitivity. Our work suggests that these problems reflect the ability of gluten peptides to inhibit cysteine uptake, perhaps contributing to chronic inflammation, although we have more to learn about that. Of course, not everybody who eats dairy or wheat has poor antioxidant capacity, and milk and wheat are important sources of nutrition. We believe that there are probably genetic vulnerabilities that bring some people closer to a critical point for oxidative stress, while for others it is a non-issue. Overall, though, this is an issue to consider in any chronic inflammatory disease or neuro-immune disease.
Methylation and glutathione are very tightly intertwined. There is a critical metabolic intersection—a fork in the road—where cells must decide to either make more glutathione, or support more methylation. The overall balance between these two options is crucial to health.
Focus: Back to glutathione. Is there another way to make it besides cysteine from the diet?
RD: Yes, your body can take homocysteine and convert it back to cysteine. Homocysteine is a metabolite of the essential amino acid methionine, and elevated levels have been associated with vascular disease. Homocysteine is created when methionine donates its methyl group to another molecule in a process known as methylation.
Methylation is a fundamental process of life which is intimately linked to redox status. In chemistry, a methyl group is a hydrocarbon molecule, or CH3. When a substance is methylated, it means that a CH3 molecule has been added to it. Methylation can regulate gene expression, protein function, even RNA metabolism. It can suppress viruses, even latent viruses or cancer viruses we are born with and can help us handle heavy metals. In the liver in particular, methylating a toxin helps change it to a form of the compound that can be more easily processed and excreted.
Methylation is an extremely broad and fundamental action that nature uses to regulate all kinds of processes. I am most fascinated by the way it regulates epigenetic changes—changes to gene expression that occur because of environmental factors—by affecting how DNA unravels during development. Some changes can be permanent for the whole lifespan and can even be passed down as many as three generations. That shows that the environment, through the process of methylation, can be quite a profound influence. There are 150-200 methyl transferase enzymes, and each enzyme can methylate multiple targets. So you can imagine methylation as a spider’s web within each cell, and that web branches out in many directions.
Methylation and glutathione are very tightly intertwined. There is a critical metabolic intersection—a fork in the road—where cells must decide to either make more glutathione, or support more methylation. The overall balance between these two options is crucial to health, and this occurs with homocysteine. When methionine gives away its methyl group, we’re left with homocysteine. And the body has to decide, should homocysteine be methylated, and go back into methionine, or should it be converted into cysteine, so that the body can make more of the antioxidant glutathione? This fundamental decision is made again and again by the body, and the overall balance is crucial to health. Too little glutathione and we will end up with free radical, oxidative damage. Not enough methylation, and many genes and viruses will not be properly regulated. Excess homocysteine, and the risk of vascular disease goes up.
It’s important to understand that multiple factors impinge on the same system. What’s so important here is that the glutathione antioxidant system is a common target for so many different environmental toxins and infections. Every single one of them impinges on the glutathione system. It’s not that each molecule of mercury or lead picks off one glutathione molecule. No. It’s that in general, environmental assaults inhibit the enzymes that are responsible for keeping the glutathione in its reduced antioxidant state, where it can do its job. The potent ability of mercury to inhibit selenium-containing enzymes is a good example.
Focus: Obviously, some people sail through these stressors and remain healthy, while others stumble and fall. Are there nutrients that more vulnerable people might find useful to supplement to help shore up the methylation/glutathione process?
RD: Though many molecules and nutrients are important, the active forms of vitamin B12 (adenosyl B12 and methyl B12) and the active form of folate (methylfolate) are essential to this process. Once you have the raw material to make glutathione or to methylate, you need cofactors like methylfolate and methylb12 to complete the process. If we don’t make enough of these active forms, we will not be able to smoothly and fluidly shift between methylation and glutathione.
Nature allows, and even encourages, genetic variation, and the bottom line is that some people have genetic variations that render this process less functional. Even with a less functional genetic legacy, you might be fine if you are not stressed by the environment—in particular by chronic infections or toxic assaults. Stress brings out limitations in genes that otherwise are latent and not problematic. That’s a general truth. So yes, with proper testing by a doctor to see if there is a functional deficiency, supplementation with active forms can help. For example, there is a test that measures levels of methylmalonic acid (MMA) in the urine; if the levels are high, you are not making enough of the two active forms of B12. Your serum B12 may be perfectly normal—you just aren’t converting enough of it to the active form.
Focus: Tell us about B12. What do the active forms of B12 do for methylation and glutathione synthesis?
RD: First, I should point out that we ourselves cannot make B12, also known as cobalamin. Bacteria make it for us, and since vegetables don’t carry those bacteria, vegans can be deficient in B12. B12 is such a precious material for the body that if, for instance, you eat a piece of rib eye steak, the B12 released from the proteins is instantly bound right there in the GI tract and chaperoned as if in a football handoff to be carried to cells, transported inside and then processed into the two active forms. Nature knows this is a precious material for life, and a critical indicator of cellular oxidation status.
There are several natural forms of B12 which need to be converted into the active forms, adenosylB12 and methylB12. CyanoB12, the form in most vitamin supplements, is not active and is less useful than the active forms for treating deficiency states. Glutathione itself is needed for converting other forms of B12 to the active forms. Indeed, there is a type of cobalamin called glutathionylcobalamin that is an intermediate for making the active forms.
There are two enzymes in the human body that require active B12 as a cofactor. One is called methylmalonyl CoA mutase, and it needs adenosyl B12. It is an enzyme that is necessary for the mitochondria—the energy powerhouse of your cell—to function. The other enzyme that requires active B12 is the enzyme methionine synthase, which requires methyl B12.
Methyl B12 is constantly recycled. It donates its methyl group to homocysteine, which then turns into methionine. Once B12 is missing its methyl group, it needs to get a fresh one. And that’s where methylfolate comes in. Methylfolate is in essence a methyl donor for methionine synthase. That’s its job in life. It is the only molecule than can donate a methyl group to B12. Once it does that, the rest of the folate is available to go out and support all kinds of other reactions in the body that need plain folate.
The glutathione antioxidant system is a common target for so many different environmental toxins. Every single one of them impinges on the glutathione system, and their effects are additive.
Focus: Tell us what happens when you don’t have enough methylB12.
RD: When your level of methylB12 is low, homocysteine builds up and this can have adverse health effects. High homocysteine levels in the blood reflect low activity of the enzyme methionine synthase, and this has been linked to an increased risk of atherosclerosis and coronary artery disease. It is also well known that homocysteine levels are increased in Alzheimer’s disease, which suggests a role for impaired methylation in this neurodegenerative disorder. Of course low B12 levels are classically associated with pernicious anemia and with peripheral neuropathy.
Focus: And what happens when you don’t have enough methylfolate?
RD: Low levels of folate are also classically associated with anemia, heart disease, fetal abnormalities such as spina bifida, as well as neuropathies and these have been specifically linked to a deficiency in methylfolate. In addition, recognition of the important role of methylfolate and vitamin B12 in supporting D4 dopamine receptor methylation links their deficiency to impaired attention such as attention-deficit hyperactivity disorder (ADHD). People with genetic polymorphisms in the enzyme that makes methylfolate are particularly vulnerable to a deficiency.
Focus: There has been some research showing that synthetic folic acid can build up when supplemented, and a few studies have suggested this may even be linked to cancer in high doses. Any thoughts about methylfolate supplementation?
RD: I’m a research scientist, not a doctor, but my private opinion is that interventions are best based on tests showing low levels. That is a sound approach for any kind of supplementation.
Focus: What about other nutrients in addition to these important B vitamins?
RD: In addition to vitamin B12 and methylfolate, there are several other nutritional supplements whose actions are critical for redox and methylation pathways. Vitamin B6 (pyridoxal-5-phosphate or P5P) is an essential cofactor for the two enzymes that sequentially convert homocysteine to cysteine, namely cystathionine-beta-synthase and cystathionine-gamma-lyase. Together these two B6-dependent enzymes comprise the transsulfuration pathway that promotes glutathione synthesis. The common supplement form of vitamin B6, pyridoxine, must be converted to the active form, and in some disorders, such as autism, this conversion is impaired, so the P5P form may be more effective. N-acetylcysteine (NAC) provides a supplementary source of cysteine. NAC can cross into the cell cytoplasm where the cysteine is released and allowed to promote glutathione synthesis. SAMe is an active, methyl-donating derivative of the essential amino acid methionine, and during oxidative conditions its levels may be low, due to low methionine synthase activity. SAMe has shown particular benefit in treating depression.
Focus: So, to go back to your initial theme, wearing redox glasses has changed your life. Any inspiring thoughts on future directions?
RD: In my opinion these examples of the interrelationship between oxidation and methylation are just the tip of the redox iceberg. Nature has learned to harness the power of oxidation as a signaling mechanism to control cellular activity. When more antioxidant is made available, cells can safely undertake a higher level of metabolic activity. There is a lot more to learn, and the real challenge will be to convert this evolving knowledge about redox and methylation into new, more effective treatment strategies.
A representative sample of Richard Deth, Ph.D.’s publications:
- Deth, R.C. “Molecular Origins of Attention: The Dopamine-Folate Connection” Kluwer Academic Publishers (April, 2003)
- Zhao R, Chen Y, Tan W, Waly M, Malewicz B, Stover P, Rosowsky A, Deth RC. Relationship between dopamine-stimulated phospholipid methylation and the single-carbon folate pathway. J Neurochem. 2001 Aug;78(4):788-96. PMID: 11520899
- Sharma A, Waly M, Deth RC. Protein kinase C regulates dopamine D4 receptor-mediated phospholipid methylation. Eur J Pharmacol. 2001 Sep 14;427(2):83-90. PMID: 11557258
- Deth RC, Sharma A, Waly M. Dopamine-stimulated solid-state signaling: A novel role for single-carbon folates in human attention. In: Proc. 12th Int. Symp. Chem. Pteridines and Folates. Kluwer Academic Press (2002).
- Zhu Q, Qi LJ, Abou-Samra A, Shi A, Deth RC. Protein kinase C regulates alpha(2A/D)-adrenoceptor constitutive activity. Pharmacology. 2004 Jun;71(2):80-90. PMID: 15118347
- Waly M, Banerjee R, Choi SW, Mason J, Benzecry J, Power-Charnitsky VA, Deth RC. Activation of methionine synthase by insulin-like growth factor-1 and dopamine: a target for neurodevelopmental toxins and thimerosal. Mol Psychiatry. 2004 Apr;9(4):358-70. PMID: 14745455
- Deth RC, Kuznetsova A, Waly M. “Attention-related signaling activities of the D4 dopamine receptor” in Cognitive Neuroscience of Attention, Michael Posner Ed., Guilford Publications Inc., New York (2004). p 269-282.
- Culley DJ, Raghavan SV, Waly M, Baxter MG, Yukhananov R, Deth RC, Crosby G. Nitrous oxide decreases cortical methionine synthase transiently but produces lasting memory impairment in aged rats. Anesth Analg. 2007 Jul;105(1):83-8. PMID: 17578961
- Kuznetsova AY, Deth RC. A model for gamma oscillations induced by D4 dopamine receptor-mediated phospholipid methylation. J Comput Neurosci. 2008 Jun;24(3):314-29. PMID: 17929154
- Deth R, Muratore C, Benzecry J, Power-Charnitsky V, Waly M. How environmental and genetic factors combine to cause autism: A Redox/Methylation Hypothesis. Neurotoxicology. 2008 Jan;29(1):190-201. PMID: 18031821
- Deth RC, Muratore C, Waly M. “Oxidative stress in autism and its implications for dopamine-stimulated phospholipid methylation” in Neurochemical Basis of Autism: Molecules to Minicolumns, Gene Blatt Ed., Springer, New York (2009).
- Kane P, Cartaxo A, Deth RC. “Nutritional issues in the causation and treatment of autism” in Food and Nutrients in Disease Management, Ingrid Kohlstadt Ed., CRC Press, Boca Raton (2008).
- Waly M, Muratore C, Power-Charnitsky V, Bojkovic J, Thomas E, Sharma A, Audhya T, Deth RC. Exon skipping in human neuronal methionine synthase associated with glutathione dependence and potent inhibition by heavy metals. PLoS One (Under review)
- Muratore C, Abdolmaleky HM, Persico AM, De La Monte S, Deth RC. Decreased methionine synthase mRNA in human cortex across the lifespan and in autism. PLoS One (Under review)
- Deth RC. “The Redox/Methylation Hypothesis of Autism: A Molecular Mechanism for Heavy Metal-induced Neurotoxicity” in Autism: Oxidative Stress, Inflammation and Immune Abnormalities, A. Chauhan, V. Chauhan, W.T. Brown Eds., Taylor & Francis/CRC Press, Boca Raton (2009).
- Mutter J, Curth A, Naumann J, Deth R, Walach H. Does Inorganic Mercury Play a Role in Alzheimer’s Disease? A Systematic Review and an Integrated Molecular Mechanism. J Alzheimers Dis. 2010;22(2):357-74. PMID: 20847438
- Terhune T, Deth R. Does vaccine-derived aluminum promote atopy via IL-18 upregulation?. Immunology and Cell Biology (Under review).
- Deth RC, Waly M, Muratore C, Hodgson N. “Redox Imbalance and the Metabolic Pathology of Autism” in Developmental Neurotoxicology Research: Principles, Models, Techniques, Strategies and Mechanisms Section V: Autism Spectrum Disorders Section, Isaac Pessah, Ed., John Wiley and Sons, Hoboken, (2010).
- Waly MI, Kharbanda KK, Deth RC. Ethanol lowers glutathione in rat liver and brain and inhibits methionine synthase in a cobalamin-dependent manner. Alcohol Clin Exp Res. 2011 Feb;35(2):277-83. PMID: 21121936
- Deth RC. “The Redox/Methylation Hypothesis of Autism”, U.S. Psychiatry 3: 48-52 (2010).
- Al-Farsi YM, Al-Sharbati MM, Waly MI, Al-Farsi OA, Al-Shafaee MA, Deth RC. “Malnutrition among preschool-aged autistic children in Oman,” in Autism Spectrum Disorders.” (2011) In Press. Epub. March26, 2011.







1 Comment. Leave new
A very informative and wonderful article as I am also putting in my efforts in understanding and treating autistic and ADHD children.