2000 years ago on the coast of Tuscany, an eighteen year old girl died and her entombed body was carefully adorned with bronze and gold jewelry. She was one of the first known sufferers of coeliac disease, an autoimmune disorder triggered by ingestion of gluten. In spite of a healthy diet of fish, meat and vegetables—which researchers determined by analyzing isotopes in her skeleton—she was only four feet seven inches tall, and many of her bones were eroded at the tips. DNA analysis revealed she carried two copies of a gene commonly found in sufferers of coeliac, and her skeletal abnormalities indicated the kind of severe malnutrition that can accompany the condition. “She probably didn’t understand that she had this disease,” Gabriele Scorrano told Nature News magazine in 2014. Scorrano is the biological anthropologist at the University of Rome who published a study on the Italian woman’s remains.1,2
2000 years ago on the coast of Tuscany, an eighteen year old girl died and her entombed body was carefully adorned with bronze and gold jewelry. She was one of the first known sufferers of coeliac disease, an autoimmune disorder triggered by ingestion of gluten. In spite of a healthy diet of fish, meat and vegetables—which researchers determined by analyzing isotopes in her skeleton—she was only four feet seven inches tall, and many of her bones were eroded at the tips. DNA analysis revealed she carried two copies of a gene commonly found in sufferers of coeliac, and her skeletal abnormalities indicated the kind of severe malnutrition that can accompany the condition. “She probably didn’t understand that she had this disease,” Gabriele Scorrano told Nature News magazine in 2014. Scorrano is the biological anthropologist at the University of Rome who published a study on the Italian woman’s remains.1,2
Two thousand years later, global knowledge about the spectrum of gluten intolerances—from genuine autoimmune disease, to gluten sensitivity, to wheat allergy—is truly state of the art. Not only can coeliac disease be determined by a suite of sophisticated antibody tests, genetic tests and monitored by in-depth analysis of biopsies, but the separate condition of gluten sensitivity can at least to some degree be tested for and monitored via symptom diaries, tests for related disorders, and elimination diets. For instance, there is an association between reported non-coeliac gluten sensitivity and irritable bowel syndrome (IBS). Among adults with IBS, up to 25 percent report clinical improvement of symptoms in response to withdrawal of gluten from the diet.3 However, some of those patients may be symptomatic from fermentable poorly absorbed, short-chain carbohydrates.4 Unfortunately, neither antigliadin antibodies nor other biomarkers can reliably identify patients with apparent non-coeliac gluten sensitivity.5,6
Nearly a third of all adults in America now want to avoid gluten in their diet.
Treatment is far less onerous than even just a few years ago, since gluten free breads, pastas, pizzas, cereals and desserts abound—all required by law to contain no more than 20 parts per million per serving (in the United States).7 Gluten free menus and clearly labeled gluten free foods are common and the gluten free food market is now estimated at over $10 billion.8,9 Customers ordered more than two hundred million dishes last year that were gluten-or wheat-free, and sales of gluten-free products are projected to surpass fifteen billion dollars by 2016, twice the amount of 2011.10
Even as gluten free foods, recipes and cookbooks become widely available, coeliac’s mysteries—such as cases that seemed to persist in spite of a gluten free diet—are now easier to understand and treat.11,12 The astonishing variety of non-gastrointestinal symptoms and conditions associated with both coeliac disease and gluten intolerance are now being recognized with new clarity.13 And new options that were once only distant gleams upon the horizon—such as digestive enzymes to dissolve and degrade gluten to even smaller gliadin peptides, or a vaccine and medications to reduce the damage from coeliac disease—are either already available or being investigated.13,14
We could almost say it’s the golden age of gluten free living, and had Miss Tuscany of 2000 years ago been born in 2015, she would likely be healthy and thriving.
Gluten: The Health Issue for Today
Today, at least seventy million Americans suffer from a digestive ailment.15 Their troubles range from bloating and abdominal distress, to gastroesophageal reflux disease (GERD), hemorrhoids, gallstones, peptic ulcers, hernias and more serious immune based inflammatory conditions like Crohn’s disease and coeliac disease.16 Nearly 1.8 million Americans have been diagnosed with celiac disease, a serious autoimmune condition triggered by gluten in grains like wheat, rye and barley. But the number of undiagnosed cases may be higher. A large-scale screening of healthy individuals in the US found that as many as one in every 105 individuals may have coeliac. A 2001 Finnish study of 3,654 children found 1 in every 99 children suffered from coeliac and 1 in 67 had coeliac antibodies as well as genetics correlated with the disease.17,18,19
Coeliac disease is a true clinical chameleon. “Ninety-plus percent of the people with the disease are not diagnosed,” says Alessio Fasano, MD.
Definitions of the disease, as well as state of the art testing for it have evolved, but one thing is certain: classical presentations of coeliac such as diarrhea, malabsorption, fatty stools and weight loss have declined since 2000, while symptoms such as bloating, gastroesophageal reflux, osteoporosis, and recurrent miscarriages, have sharply increased.20,21 In addition, recommendations for screening are evolving, as is recognition of coeliac sufferers’ heightened risk for numerous autoimmune conditions, chronic disease manifestations and even cancers.
Aside from coeliac, there is a flurry of new research into the etiology and symptoms of gluten sensitivity—an increasingly prevalent condition which, though potentially partially immune mediated, is not a true autoimmune disease like coeliac. Our understanding of this emerging disorder is still in its infancy, but we do know that with its myriad of “extra-intestinal” symptoms such as fatigue and fogginess, gluten sensitivity may already affect as many as 18 million Americans.22 An astonishing 30% of adults in America now want to avoid gluten in their diet, according to the Dieting Monitor tracking service of the NPD Group, a firm that compiles global data in more than 20 industries.23 NPD calls gluten the health issue of the current day.24
A Clinical Chameleon: The Nuanced New View of Celiac
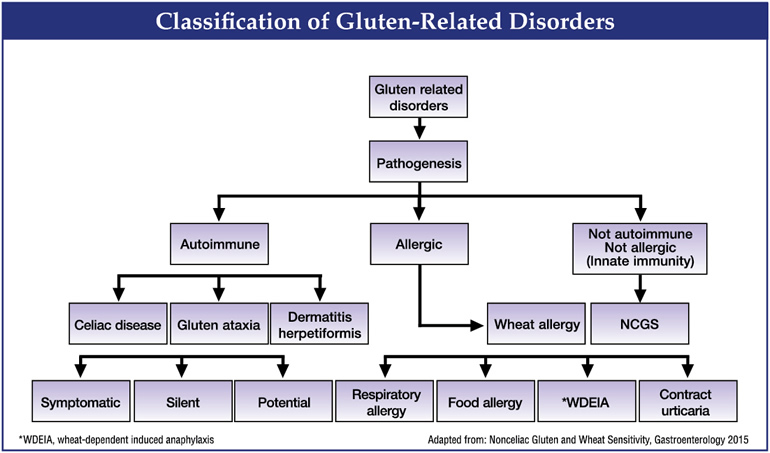
Once thought to be rare, coeliac disease (CD) is now widely recognized as a unique autoimmune disease triggered in genetically susceptible individuals by gluten, which is present in grains like wheat, rye, and barley. The pathogenesis of celiac disease involves a triad of predisposing genes (HLA-DQ2 and HLA-DQ8 haplotypes), dietary gluten, and other less well defined environmental factors. Innate and adaptive immune responses to gluten fragments at the small intestinal epithelium result in characteristic autoantibodies (esp. transglutaminase 2 that binds to gluten), histologic changes (intraepithelial lymphocytosis and villous atrophy), and various clinical symptoms. More than 95% of individuals have one of two haplotypes: HLA-DQ2 or DQ8.25
In coeliac, gluten triggers an immune response that ignites widespread tissue damage—first and foremost, atrophy of the brush-like villi, which are tiny, finger-like projections that protrude from the epithelial lining of the intestinal wall. Villi vastly increase the surface area of the small intestine, allowing it to efficiently absorb nutrients. Removal of gluten in most cases allows the restoration of villi to their healthy state and functionally “cures” coeliac. The classic symptoms of coeliac are diarrhea, malabsorption, fatty stools and villous atrophy, seen on biopsy.26
What other autoimmune disease could be so simple—a known cause, that when removed, cures the disease? But celiac is actually a far more complex illness than was originally determined. Pioneering researcher Alessio Fasano, MD, who heads a multidisciplinary team at the Center for Coeliac Research at Massachusetts General Hospital, calls the condition a true clinical chameleon.27 According to Fasano, the Center for Disease Control now considers coeliac disease a public health threat because so many sufferers go unrecognized and misdiagnosed. “Ninety-plus percent of the people with the disease are not diagnosed.”19
Coeliac does not just damage the villi. When the immune system attacks gluten, it generates autoantibodies to one of its own enzymes, called tTG (tissue transglutaminase). This enzyme gets targeted simply because it binds to gluten in the lining of the intestines. Unfortunately, tTG is found not only in the gut, but the skin, heart, thyroid, bones, and nervous system. That’s why coeliac disease can trigger damage in any of those tissues. “Name an organ, and it can be affected,” says Dr. Peter Green, director of the Coeliac Disease Center at Columbia University’s College of Physicians and Surgeons.28 That renders a very confusing clinical picture that includes anemia, abdominal symptoms similar to irritable bowel syndrome (IBS), neuropathy, ataxia, balance problems, depression, anxiety, dental enamel defects, osteoporosis, liver disease, delayed puberty, missed menstrual periods, infertility, canker sores, skin rashes, fatigue, and headaches.10 The National Foundation for Coeliac Awareness offers an online checklist of possible symptoms.29
The coeliac chameleon also shows up in clinically “silent” but present disease. Even in Finland, where the level of awareness of the disease is high and the most sensitive and specific blood tests are available along with genetic typing, silent cases are missed.16 In one striking Finnish study, blood samples from 3,654 students (aged seven to sixteen years old) were collected in 1994, and later screened for coeliac. Fifty-six had positive antibody tests (1.5%), but only ten had been diagnosed by 2001. Another 27 in the study turned out to have coeliac disease on biopsy.30
Silent Is Not Benevolent
Silent does not mean benevolent: undiagnosed coeliac cases carry a risk of long-term complications, including osteoporosis, infertility, and T-cell lymphoma. Healthy adults at an Air Force base in Minnesota had blood drawn and stored between 1948 and 1954; testing showed that 14 (0.2 percent) had undiagnosed coeliac disease. At a follow-up 45 years later, these same individuals had a fourfold increase in the Kaplan-Meier all-cause mortality rate compared to their non-coeliac peers, and adjusted for age, sex, and enlistment status. (Note: the widely used Kaplan-Meier estimate is the simplest way of computing survival rate over time.)31
Zonulin, a molecule that modulates intestinal permeability, can be thirty times higher in individuals with coeliac disease—even when they are on a gluten free diet—than individuals without the disease.
Even clinically silent coeliac disease benefits from a gluten free diet. In a 2014 study, over 3,000 individuals were screened for coeliac and 40 were chosen who had positive blood tests for coeliac antibodies. They were put on either a gluten containing or gluten free diet and were followed for a year. The height of the villi on those individuals following a gluten free diet increased, while their levels of coeliac antibodies decreased. Nonspecific gastrointestinal symptoms improved as well (including indigestion and reflux).32
Not only is the clinical picture of coeliac changing, its incidence is rising sharply. This is a mystery nobody has yet truly deciphered—what exactly about gluten-containing grains, or the environment has changed to increase the number of cases? We have been eating wheat, and thus gluten, for ten thousand years. What has changed? Known cases have nearly quadrupled since 1950.33 Even since 1994, the increase has been sharp: testing of over 17,000 adults revealed 0.17% of adults aged 50 years and older had coeliac between 1988 and 1994 while 0.44% had it between 2009 and 2012.34
Does an Altered Gut Flora Break Gluten Tolerance?
Chameleon coeliac can strike suddenly, at any time in a person’s life, suggesting that tolerance to gluten can be abruptly and mysteriously broken—perhaps by life stresses such as infection or even imbalances in the gut microbiome and flora. An inflammatory immune molecule, interleukin 17 (IL-17), which can contribute to chronic gut inflammation and dysbiosis, has been found in a subset of coeliac sufferers.35 And astonishing research by Anthony Fasano uncovered a protein called zonulin that modulates the intestinal permeability—by regulating what are called tight junctions between cells of the wall of the digestive tract. With his team at the Massachusetts General Hospital, Fasano discovered the molecule in 2000, and in 2006 they found that zonulin can be thirty times higher in individuals with coeliac disease—even when they are on a gluten free diet for two years—than individuals without the disease.36,37 In other words, their gluten-free guts are three times leakier than those who don’t have coeliac. In addition, groundbreaking new research finds that individuals with non-celiac gluten sensitivity also have elevated levels of zonulin. At the 23rd United European Gastroenterology Week (UEG Week, 2015) in Barcelona, Spain, Giovanni Barbara from the University of Bologna noted that: “Blood levels of zonulin were almost as high in patients with NCGS (Non-Coeliac Gluten Sensitivity) as in those with coeliac disease.”
Coeliac disease can trigger damage in almost any tissue. “Name an organ, and it can be affected,” says Dr. Peter Green, director of the Coeliac Disease Center at Columbia University’s College of Physicians and Surgeons.
Now here’s a fascinating clue for a disease detective: intestinal bacteria such as E. coli and Salmonella stimulate zonulin production and increase the ability of gluten to cause intestinal damage. Researchers speculate that this zonulin-driven opening may help flush out microorganisms and spur the body to fight bacterial colonization of the small intestine.38 But chronic upregulation could be harmful. Rod shaped bacteria have been found in the mucosa of individuals with CD, both inactive and active disease, but not in controls.60 Research shows that the development of the gut microbiome and its composition may play an active role in the pathogenesis of CD.39 In the laboratory, inoculating intestinal tissue with healthy lactobacilli and bifidus protects the intestinal lining from gluten-induced damage.40,41 We are also in the very early phase of exploring the gut virome—viruses that interact with the gut’s immune system and flora—and may contribute to ongoing inflammation.42 Immunologists and microbiologists are working on the notion that one cause for the increase in coeliac—or its appearance later in life—could be intestinal dysbiosis.43,44,45,46,47
There is more than our internal milieu which may orchestrate our vulnerability to coeliac disease. The milieu we grow up in—and the healthy diversity of microorganisms around us—may also be important. One fascinating study of 5,500 individuals living in two nearby towns, one in modern day Finland, the other across the border in a more rural and traditional Russian environment, found remarkably different rates of coeliac. One in every 100 Finnish children had coeliac, but only one in every 500 Russian children succumbed to the disease. Differing wheat consumption patterns were irrelevant—if anything, Russians consume more wheat than Finns, and similar types. The difference seemed to be in the environment—and the apparently healthy range of microorganisms children were exposed to. Or, as the researchers wryly put it, “This [effect] may be associated with a protective environment characterized by inferior prosperity and standard of hygiene in Karelia, Russia.48 This has bearing on the now widely known hygiene hypothesis, which proposes that a lack of childhood exposure to infectious agents, microorganisms such as those normally in the gut flora or probiotics, and parasites increases susceptibility to allergic diseases by suppressing the natural development of the immune system.49,50
As Alessio Fasano said in a 2010 interview, “Let’s say all of a sudden you get an infection, you take antibiotics, you have surgery, you have a pregnancy, which by the way are all examples in which your bacteria [microbiome] will change tremendously…a lot of cases of coeliac disease start after one of these occur. What happened now, you have another microbiome, another set of genes from new bacteria, that switches [you] from tolerance to gluten to the autoimmune process. That is the only way I can explain these results where people who, despite having the genes, did not have coeliac disease (as determined by positive blood tests and other measures) until they were 60 years old.”19
The New Elephant in the Room: Non-Celiac Gluten Sensitivity
Move over coeliacs, you have company: 18 million Americans may be gluten intolerant (caveat: this is simply an estimate, since many of these individuals started a gluten free diet [GFD] without medical advice or consultation.)51 They are loosely grouped under the name Non-Coeliac Gluten Sensitivity (NCGS), which was first described as far back as the 1980s but more finely characterized and separated from coeliac in 2010.52 This is not an autoimmune disease, and it does not damage intestinal villi the way that coeliac disease does, nor is it a precursor for coeliac development. When biopsied, individuals with NCGS revealed normal to mildly inflamed mucosa, while celiac patients showed partial villous atrophy.43 NCGS shows up as symptoms that occur soon after gluten ingestion, disappear with gluten withdrawal and relapse following gluten challenge, within hours or days. The “classical” presentation of NCGS is a combination of IBS-like symptoms, including abdominal pain, bloating, diarrhea or constipation, and systemic manifestations such as brain fog, headache, fatigue, joint and muscle pain, leg or arm numbness, dermatitis (eczema or skin rash), depression, and even anemia.53
18 million Americans may be gluten intolerant, and suffer from Non-Coeliac Gluten Sensitivity.
The diagnosis of NCGS is based on the suspicion of hyper-reactivity to gluten and the absence of coeliac disease. Could this be due to a placebo effect? No, according to a double-blind randomized placebo-controlled study from 2012. IBS-like symptoms of NCGS were more frequent in a group given gluten (68%) than in those given a placebo (40%).54 And there were no significant differences between CD and NCGS patients regarding personality traits, level of somatisation, quality of life, anxiety, and depressive symptoms. Symptoms upon gluten challenge were not related to personality in NCGS patients.55
New research shows that individuals who suffer from IBS that manifests mainly as bouts of diarrhea (IBS-D) may indeed be gluten sensitive, according to new research. These individuals have leakier guts on a gluten containing diet (GCD), and researchers have concluded that gluten can alter bowel barrier functions in patients with IBS-D.55,56
When Gluten is Not At Fault: Fermentable Carbohydrates Or Wheat Allergy?
However, the science of gluten sensitivity is still evolving. In one renowned 2011 study from Australia, 34 individuals with IBS were put on a gluten-free diet, but half the subjects got muffins and bread with gluten. Neither the patients nor doctors knew who was ingesting gluten, so the well-designed study was double-blind. The majority on gluten reported that the pain returned.57
According to Mayo Clinic’s Joseph R. Murray, that study “blew our minds…when people were challenged with gluten, by eating the muffins, they got sick.”7 But then the same researchers carried out a second study, recruiting 37 seemingly gluten-intolerant individuals. They were put on a gluten-free diet known as FODMAPS that was also free of carbohydrates (fermentable oligosaccharides, disaccharides, monosaccharides, and polyols). FODMAPS are common in foods ranging from dairy products to fruit. Some individuals experience abdominal pain, bloating, and diarrhea after ingesting FODMAPS. In the second study, individuals on a gluten and FODMAPS free diet felt better within two weeks. When they were then secretly given gluten-containing foods that did not harbor FODMAPS, they did not relapse.58
Another big contender in wheat sensitivity has nothing to do with gluten. It is a class of molecules called amylase trypsin inhibitors (ATIs). They are potent stimulators of the immune system, and can cause immune system cells to release inflammatory molecules, including interleukin-8 (IL-8) and tumor necrosis factor-alpha (TNF-a).
The takeaway? Though non-celiac gluten sensitivity is a real phenomenon, there can be other components of wheat, rye and barley products that cause symptoms in sensitive individuals. Self-diagnosis is not always reliable, and there is also the incidence of wheat allergy to consider. Wheat allergy is a bonafide allergic reaction to foods containing wheat, one of the top eight food allergens in the United States.60 Symptoms can include swelling, itching or irritation of the mouth or throat, hives, headaches, itchy and watery eyes, cramps, nausea, vomiting, diarrhea and more. Wheat allergy is most common in babies and toddlers, who often outgrow it as their digestive and immune systems mature. Adults can also develop wheat allergy, sometimes as a cross-sensitivity to grass pollen and it is characterized by an IgE related immune response.61
Gluten-Free but Glutened Anyway?
A gluten free diet is simple in concept, profound in its life-changing ability to cure coeliac, but extremely challenging for many to follow. The confections made of wheat—those delectable loaves of bread, the buttery crust of pies, the oh-so-satisfying crunch of crisp crackers—are everywhere to tempt us, and wheat’s amber waves of grain grow worldwide (not to mention other gluten-containing grains such as barley and rye). In 2013, the world harvested seven hundred and eighteen million tons of wheat. Moreover, avoiding the obvious is not enough. Gluten and even wheat can be hidden in commercial soups, gravies, processed meats, sauces, and many other foods. Gluten can also contaminate gluten-free foods if they are processed in the same room or by the same machinery at a facility that isn’t properly cleaned.
Of over 5,000 Canadians with biopsy-confirmed coeliac disease surveyed, 18.8% said they had intentionally consumed gluten once or twice during the previous year. Another 13.2% said they had deliberately eaten gluten at least once a month during the previous year.
Many patients regard a gluten-free diet as a substantial burden even after being on the diet for several years.62 It’s no wonder even the most resolutely gluten-free folks inadvertently eat gluten-containing foods—or even deliberately expose themselves to the temptations of gluten. Of over 5,000 Canadians with biopsy-confirmed coeliac disease surveyed, 18.8% said they had intentionally consumed gluten once or twice during the previous year. Another 13.2% said they had deliberately eaten gluten at least once a month during the previous year.63 That’s an alarming number of coeliac sufferers who are knowingly harming their health to indulge in the seemingly irresistible temptation of glutenous goods. These figures don’t account for the thousands of other gluten sensitive and intolerant individuals that continue to consume gluten-containing foods.
But even those who think they are conscientiously living a gluten-free life might be mistaken. When asked to review a list of 15 foods and ingredients and identify those that were not allowed on a gluten-free diet, only half of respondents in the above study correctly identified all seven non-allowed items. A third of the coeliacs surveyed thought imitation crab meat, often found in sushi dishes, was safe.63 Inadvertent gluten exposure may explain the fact that at least 30% of coeliac sufferers on a gluten-free diet simply do not fully recover.64 Mayo Clinic’s Dr. Joseph Murray and his colleagues reported that of 381 adults with biopsy-proven coeliac disease, 34% had full mucosal recovery after 2 years on a gluten-free diet. After 5 gluten-free years, the percentage jumped to 66%. But 34% still had active disease—and greater risk of all-cause mortality.65 In an impressive 2013 study, Dr. Alessio Fasano followed 17 patients who did not respond to a gluten-free diet. They had persistent symptoms and continued villous atrophy. They were considered to suffer from non-responsive coeliac disease (NRCD). But when Fasano put all 17 of those patients on a highly restricted diet that would eliminate even trace gluten, 14 responded. Eleven of those 14 healed enough to go back to a regular gluten-free diet. Fasano’s strict diet prohibits all cereal grains except rice, and focuses on fruits, vegetables and herbs, fresh meats, poultry, fish and unflavored, unseasoned dairy products.66
As of 2013, a gluten free product in America or Europe must contain less than 20 parts per million (ppm) per serving. In Australia and New Zealand, the cutoff is 5 ppm. Europe has added a category between 20 and 100 ppm called “very low gluten” for gluten intolerant individuals. In Canada, gluten-free indicates that a food does not contain wheat, spelt, kamut, rye, barley, oats or triticale, or any parts of those grains.67
Are these levels safe? Studies have shown that coeliac sufferers eating 200 mg of gluten daily will have villous atrophy within four weeks. An often-cited 2007 Italian study followed 49 coeliac sufferers who were given capsules containing either zero, 10 or 50 mg of gluten for three months. Fifty mg of gluten a day caused significant villous atrophy. That has become the unofficial “cutoff” for safety guidelines, but in that study, one coeliac sufferer consuming only 10 mg of gluten daily had a relapse.68,69 Given today’s gluten-free labeling, ten ounces of “gluten-free” food containing 20 ppm would contain 5.7 mg of gluten.70 Total daily portions are important to keep in mind if one is eating a lot of gluten-free products; however, most gluten-free food servings probably contain less than 20 ppm.
A Little Extra Help From Tailored Enzymes
Today, a gluten-free diet is the only medically accepted treatment for celiac disease. And for the gluten intolerant, it keeps symptoms at bay. Will there come a day when those with coeliac and NCGS can eat gluten happily? Can coeliac disease be functionally “cured?” Several pharmaceutical companies are in the race to develop enzyme based medicines that effectively and completely degrade gluten or inhibit zonulin.71 These developments have created tremendous excitement in the field of gastroenterology.
“If you are gluten intolerant you can benefit from enzymes that help digest and degrade gluten.” Theodore Hersh, MD.
The problem with gluten is that our own intestinal enzymes do not easily degrade gliadin –which is rich in proline and glutamine. In the genetically susceptible individual, this can trigger an immune reaction.72 In addition, coeliacs who have mucosal damage already secrete fewer intestinal digestive enzymes.73 Enzyme supplement therapy is not an alternative to the gluten-free diet—though one day it may be. But for now, it may enable patients to ease their worries about cross contamination and inadvertent exposure; and it might help lessen symptoms in the gluten sensitive. In the words of gastroenterologist Theodore Hersh, of Emory University in Atlanta, “If you are gluten intolerant, and you eat a sandwich, you can benefit from enzymes that help digest and degrade gluten.”74
An enzyme that inhibits zonulin has been tested in mice and humans, and has been found effective and well tolerated. It may prove helpful in the case of inadvertent exposure. The pharmaceutical company, Alba Pharmaceutical, has obtained permission from the Food and Drug Administration (FDA) to expand the study from just coeliac disease to other autoimmune diseases, such as type 1 diabetes or Crohn’s disease, which are characterized by increased zonulin levels.75
Enzymes that degrade and digest gluten are also being intensively studied. One commonly used enzyme is called Dipeptidyl peptidase IV (DDP IV). It is an exo-peptidase enzyme, which means it can cleave off the ends of a peptide (protein). A decrease in intestinal DPP IV activity has been shown to correlate with the level of mucosal damage in patients with celiac disease and other malabsorption syndromes.76 DDP IV can degrade gliadin, but since it only works on the end (or terminal) bonds, it is only partially effective.76 It also works best at a neutral pH, and not the acidic environment of the stomach.77 On its own, it is not powerful enough for either celiac disease nor NCGS as much of the molecule will be left intact. A mold called Aspergillus niger that produces an enzyme called aspergillopepsin has been used in conjunction with DPP IV. DPP IV markedly enhances ASP’s ability to degrade gluten; however, as stated, it is inactive at the pH of the stomach, thus researchers have suggested it would need to be used with antacids.78
Along with DDP IV, adding enzymes that can actually cleave, cut and degrade internal bonds of gliadin—enzymes called endo-peptidases—can powerfully increase effectiveness.79 These enzymes efficiently degrade gluten, turning it into small and harmless molecules, which are well tolerated in individuals with coeliac.80
A 2009 study evaluated two food-grade enzymes, aspergillopepsin (ASP) from Aspergillus niger and dipeptidyl peptidase IV (DPP IV) from Aspergillus oryzae. The ability of each enzyme to hydrolyze gluten was tested against synthetic gluten peptides, a recombinant gluten protein, and simulated gastric digestion of whole gluten and whole-wheat bread. When used alone, neither ASP nor DPP IV efficiently cleaved gluten peptides. However, when used together the blend was able to degrade moderate amounts of gluten.81 Another enzyme being studied currently is Aspergillus niger prolylendoprotease (AN-PEP). In a 2015 study, this enzyme significantly enhanced gluten digestion in the stomach of healthy volunteers.82
In one double-blind, randomized, controlled study of eleven non-coeliac gluten sensitive individuals, an enzyme containing DPP IV, pepsin derived from aspergillus, and proteases proved beneficial for gluten sensitive individuals. Eleven individuals answered a 15-item questionnaire rating digestive symptoms, health, pain and compliance with a gluten-free diet. They filled out the questionnaire before the week-long trial and then afterwards. The enzyme blend, whose complex of ingredients have been demonstrated effective at a pH ranging from 2 to 7, reduced pain in the abdominal area, rumbling of the tummy, bloating, trouble emptying bowels, hunger pains, lower energy levels, headaches and food cravings.83
A more recent double-blind, randomized, controlled study followed 37 self-reported non-coeliac gluten sensitive individuals aged 19 to 64 years of age. Twelve were on the enzyme blend, and 27 on the placebo. The enzyme blend taken three times daily for thirty days demonstrated statistically significant reduced levels of anti-gliadin IgA antibodies and C-reactive protein (a measure of inflammation), compared to those on placebo. Those on the enzymes also experienced a statistically significant reduction in reflux (or GERD), as well as food cravings.84,85
Ultimately, a comprehensive enzyme blend that effectively digests gluten, as well as other carbohydrates, fats and proteins that may often lead to sensitivities or symptoms (or for those already suffering from them), may be a beneficial lifestyle tool. If we wonder why so many with coeliac cheat and eat gluten anyway, we need to remember that a meal is a communal act that nurtures connection while literally keeping us alive. As superstar chef Lidia Bastinovich puts it, “When you sit at the table you are open to being nourished and you are more vulnerable. Food is who we are, and embedded in our foods are the flavors and aromas that take us back to our past. Food transcends beyond us. When you cook for somebody, you transport love with food. All this is so powerful.”87 Let food be our medicine, with a little help from enzyme friends when necessary.
References:
- Scorrano G,*, Brilli M, Martínez-Labarga M, Giustini F, Pacciani D, Chilleri F, Scaldaferri F, Gasbarrini A, Gasbarrini G, Rickards O. Palaeodiet reconstruction in a woman with probable celiac disease: A stable isotope analysis of bone remains from the archaeological site of Cosa (Italy) American Journal of Physical Anthropology (2014 July); 154(3): 349–56. PMID: 24706415 View Abstract
- http://www.nature.com/news/ancient-bones-show-signs-of-struggle-with-coeliac-disease-1.15128
- Biesiekierski JR, Newnham ED, Irving PM, Barrett JS, Haines M, Doecke JD, Shepherd SJ, Muir JG, Gibson PR Gluten causes gastrointestinal symptoms in subjects without celiac disease: a double-blind randomized placebo-controlled trial. Am J Gastroenterol. 2011;106(3):508. PMID 21224837 View Abstract
- Biesiekierski JR, Peters SL, Newnham ED, Rosella O, Muir JG, Gibson PR. No effects of gluten in patients with self-reported non-celiac gluten sensitivity after dietary reduction of fermentable, poorly absorbed, short-chain carbohydrates. Gastroenterology. 2013 Aug;145(2):320-8. PMID 23648697 View Abstract
- Stoven S, Murray JA, Marietta E. Celiac disease: advances in treatment via gluten modification. Clin Gastroenterol Hepatol. 2012 Aug;10(8):859-62. PMID: 22728383 View Abstract
- Husby S, Murray JA. Gluten sensitivity: celiac lite versus celiac like. J Pediatr. 2014 Mar;164(3):436-8. PMID: 24411520 View Abstract
- Thompson T.. doi: The gluten-free labeling rule: what registered dietitian nutritionists need to know to help clients with gluten-related disorders. J Acad Nutr Diet. 2015 Jan;115(1):13-6. PMID: 25534893 View Abstract
- Leonard M, Vasagari B. US Perspective On Gluten Related Diseases. Clinical and Experimental Gastroenterology 2014 Jan 24;7: 25-37. PMID: 24493932 View Abstract
- http://www.foodnavigator-usa.com/Markets/What-s-the-size-of-the-US-gluten-free-prize-490m-5bn-or-10bn
- http://www.newyorker.com/magazine/2014/11/03/grain
- Hollon JR, Cureton PA, Martin ML, Puppa EL, Fasano A. Trace gluten contamination may play a role in mucosal and clinical recovery in a subgroup of diet-adherent non-responsive celiac disease patients. BMC Gastroenterol. 2013 Feb 28;13:40. PMID: 2344840 View Abstract
- Boggs, W. Better Adherence to Gluten-Free Diet Improves Mucosal Recovery in Celiac Disease. Medscape. Jun 15, 2015
- Schuppan, D, Walburga D. Pathogenesis, epidemiology, and clinical manifestations of celiac disease in adults. UpToDate 8/2015. Accessed 9/17/2015
- http://www.dailyuw.com/science/article_5a907b9b-709b-56df-bc42-b1aa3922d18f.html
- National Institutes of Health, U.S. Department of Health and Human Services. Opportunities and Challenges in Digestive Diseases Research: Recommendations of the National Commission on Digestive Diseases. Bethesda, MD: National Institutes of Health; 2009. NIH Publication 08–6514.
- National ambulatory medical care survey: 2010 emergency department summary tables. Centers for Disease Control and Prevention website http://www.cdc.gov/nchs/data/ahcd/namcs_summary/2012_namcs_web_tables.pdf
- Lohi S, Mustalahti K, Kaukinen K, et al. Increasing prevalence of coeliac disease over time. Aliment Pharmacol Ther. 2007 Nov 1;26(9): 1217–25. PMID: 17944736
- Mäki M, Mustalahti K, Kokkonen J, Kulmala P, Haapalahti M, Karttunen T, Ilonen J, Laurila K, Dahlbom I, Hansson T, Höpfl P, Knip M. Prevalence of Celiac disease among children in Finland. N Engl J Med. 2003 Jun 19;348(25):2517-24. PMID: 12815137 View Abstract
- Ratner, A. Dr. Fasano on the Future of Celiac Disease. Gluten-Free Living, Dec. 2010, pp 40-54
- Ludvigsson JF, Rubio-Tapia A, van Dyke CT, Melton LJ 3rd, Zinsmeister AR, Lahr BD, Murray JA.Am J Gastroenterol. Increasing incidence of celiac disease in a North American population. 2013 May;108(5):818-24. PMID: 2351146 View Abstract
- Rubio-Tapia A,, Hill ID, Kelly CP , Calderwood AH, Murray, JA. ACG Clinical Guidelines: Diagnosis and Management of Celiac Disease, Amer Jnl Gastroenterology. Vol 108, May 2013, pp. 656-676. PMID: 23609613 View Abstract
- http://www.celiaccentral.org/non-celiac-gluten-sensitivity/
- https://www.npd.com/wps/portal/npd/us/about-npd/
- http://www.foodbusinessnews.net/articles/news_home/Food-Service-Retail/2013/06/Food_service_eyed_as_the_next.aspx?ID=%7B648702AA-1D1E-4715-815C-F7D39DEF8977%7D&cck=1
- Kaukinen K, Partanen J, Mäki M, Collin P. HLA-DQ typing in the diagnosis of celiac disease. Am J Gastroenterol. 2002 Mar;97(3):695-9. PMID: 11922565 View Abstract
- http://www.niddk.nih.gov/health-information/health-topics/digestive-diseases/celiac-disease/Pages/facts.aspx
- Fasano, A. Celiac Disease: How to handle a clinical chameleon. NEJM, 2003 June 19;348(25):2568-70. PMID: 12815143 1 View Abstract
- http://www.saturdayeveningpost.com/2010/03/01/in-the-magazine/health-in-the-magazine/celiac-disease-great-pretender.html
- http://www.celiaccentral.org/checklist/
- Mäki M, Mustalahti K, Kokkonen J, Kulmala P, Haapalahti M, Karttunen T, Ilonen J, Laurila K, Dahlbom I, Hansson T, Höpfl P, Knip M. Prevalence of Celiac disease among children in Finland. N Engl J Med. 2003 Jun 19;348(25):2517-24. PMID: 12815137 View Abstract
- Rubio-Tapia A, Kyle RA, Kaplan EL, et al. Increased prevalence and mortality in undiagnosed celiac disease. Gastroenterology 2009 Jul; 137(1):88-93. PMID: 19362553 View Abstract
- Kurppa K, Paavola A2 Collin P, Sievänen H, Laurila K, Huhtala H, Saavalainen P, Mäki M, Kaukinen K. Benefits of a gluten-free diet for asymptomatic patients with serologic markers of celiac disease. Gastroenterology. 2014 Sep;147(3):610-17. PMID: 24837306 View Abstract
- http://www.mayo.edu/research/discoverys-edge/celiac-disease-rise
- Choung RS, Ditah IC, Nadeau AM, Rubio-Tapia A, Marietta EV, Brantner TL, Camilleri MJ, Rajkumar SV, Landgren O, Everhart JE, Murray JA. Trends and racial/ethnic disparities in gluten-sensitive problems in the United States: findings from the National Health and Nutrition Examination Surveys from 1988 to 2012. Am J Gastroenterol. 2015 Mar;110(3):455-61. PMID: 25665935 View Abstract
- Eiró N, González-Reyes S, González L, González LO, Altadill A, Andicoechea A, Fresno-Forcelledo MF, Rodrigo-Sáez L, Vizoso FJ. Duodenal expression of Toll-like receptors and interleukins are increased in both children and adult celiac patients. Dig Dis Sci. 2012 Sep;57(9):2278-85 36
- Fasano A, Not T, Wang W, Uzzau S, Berti I, Tommasini A, Goldblum SE. Zonulin, a newly discovered modulator of intestinal permeability, and its expression in coeliac disease. Lancet. 2000 Apr 29;355(9214):1518-9. PMID: 10801176 View Abstract
- Wang W, Uzzau S, Goldblum SE, Fasano A. Human zonulin, a potential modulator of intestinal tight junctions. J Cell Sci. 2000 Dec;113 Pt 24:4435-40.
- Barbaro MR, et al. The role of zonulin in non-celiac gluten sensitivity and irritable bowel syndrome. Abstract presented at the 23rd United European Gastroenterology Week (UEG Week 2015), Oct. 24-27 2015, Barcelona, Spain. 2. Fasano A. Ann NY Acad Sci 2012;1258:25-33.
- El Asmar R, Panigrahi P, Bamford P, Berti I, Not T, Coppa GV, Catassi C, Fasano A.Host-dependent zonulin secretion causes the impairment of the small intestine barrier function after bacterial exposure. Gastroenterology. 2002 Nov;123(5):1607-15. PMID: 12404235 View Abstract
- Sanz Y, Sánchez E, Marzotto M, Calabuig M, Torriani S, Dellaglio F. Differences in faecal bacterial communities in coeliac and healthy children as detected by PCR and denaturing gradient gel electrophoresis. FEMS Immunol Med Microbiol. 2007 Dec;51(3):562–8. PMID: 17919298 View Abstract
- Orlando A, Linsalata M, Notarnicola M, Tutino V, Russo F Lactobacillus GG restoration of the gliadin induced epithelial barrier disruption: the role of cellular polyamines. BMC Microbiol, 2014 Jan 31;14:19. PMID: 24483336 View Abstract
- Lamprecht M, Bogner S, Schippinger G, Steinbauer K, Fankhauser F, Hallstroem S, Schuetz B, Greilberger JF. Probiotic supplementation affects markers of intestinal barrier, oxidation, and inflammation in trained men; a randomized, double-blinded, placebo-controlled trial. J Int Soc Sports Nutr. 2012 Sep 20;9(1):45. PMID: 22992437 View Abstract
- Focà A, Liberto MC, Quirino A, Marascio N, Zicca E, Pavia G. Gut inflammation and immunity: what is the role of the human gut virome? Mediators Inflamm. 2015;2015:326032. PMID: 2594498 View Abstract
- Fasano A. Intestinal permeability and its regulation by zonulin: diagnostic and therapeutic implications. Clin Gastroenterol Hepatol. 2012 Oct;10(10):1096-100. PMID: 22902773 View Abstract
- Fasano A. Zonulin, regulation of tight junctions, and autoimmune diseases. Ann N Y Acad Sci. 2012 Jul;1258:25-33. doi: 10.1111/j.1749-6632.2012.06538.x. PMID: 22731712 View Abstract
- Fasano A. Zonulin and its regulation of intestinal barrier function: the biological door to inflammation, autoimmunity, and cancer. Physiol Rev. 2011 Jan;91(1):151-75. PMID: 21248165 View Abstract
- Drago S, El Asmar R, Di Pierro M, Grazia Clemente M, Tripathi A, Sapone A, Thakar M, Iacono G, Carroccio A, D’Agate C, Not T, Zampini L, Catassi C, Fasano A.Scand J Gastroenterol. Gliadin, zonulin and gut permeability: Effects on celiac and non-celiac intestinal mucosa and intestinal cell lines. 2006 Apr;41(4):408-19. PMID: 16635908 View Abstract
- Wang W, Jovel J, Halloran B, Wine E, Patterson J, Ford G, O’Keefe S, Meng B, Song D, Zhang Y, Tian Z, Wasilenko ST, Rahbari M, Reza S, Mitchell T, Jordan T, Carpenter E, Madsen K, Fedorak R, Dielemann LA, Ka-Shu Wong G, Mason AL. Metagenomic analysis of microbiome in colon tissue from subjects with inflammatory bowel diseases reveals interplay of viruses and bacteria.Inflamm Bowel Dis. 2015 Jun;21(6):1419-27. PMID:25939040 View Abstract
- Kondrashova A, Mustalahti K, Kaukinen K, Viskari H, Volodicheva V, Haapala AM, Ilonen J, Knip M, Mäki M, Hyöty H; Epivir Study Group. Lower economic status and inferior hygienic environment may protect against celiac disease. Ann Med. 2008;40(3):223-31. PMID: 18382888 View Abstract
- Okada H, Kuhn C, Feillet H, Bach JF. The ‘hygiene hypothesis’ for autoimmune and allergic diseases: an update. Clin Exp Immunol. 2010 Apr;160(1):1-9. PMID: 20415844 View Abstract
- Dr. Fasano on the future of celiac disease, Gluten Free Living, 2010, pp. 40-54. www.glutenfreeliving.com
- http://www.massgeneral.org/children/services/celiac-disease/gluten-sensitivity-faq.aspx
- Sapone, A.; Lammers, K.M.; Mazzarlla, G.; Mikhailenko, I.; Cartenì, M.; Casolaro, V.; Fasano, A. Differential mucosal IL-17 expression in two gliadin-induced disorders: Gluten sensitivity and the autoimmune enteropathy celiac disease. Int. Arch. Allergy Immunol. 2010: 152, 75–80. PMID: 19940509 View Abstract
- Catassi C, et al. Non-Celiac Gluten sensitivity: the new frontier of gluten related disorders. Nutrients. 2013 Sep 26;5(10):3839-53. PMID: 24077239 View Abstract
- Biesiekierski, J.R.; Newnham, E.D.; Irving, P.M.; Barrett, J.S.; Haines, M.; Doecke, J.D.; Shepherd, S.J.; Muir, J.G.; Gibson, P.R. Gluten causes gastrointestinal symptoms in subjects without celiac disease: A double-blind randomized placebo-controlled trial. Am. J. Gastroenterol. 2011 Mar;106, 508–514. PMID: 21224837 View Abstract
- Vazquez-Roque, M.I.; Camilleri, M.; Smirk, T.; Murray, J.A.; Marietta, E.; O’Neill, J.; Carlson, P.; Lamsam, J. A controlled trial of gluten-free diet in patients with irritable bowel syndrome-diarrhea: Effects on bowel frequency and intestinal function. Gastroenterology 2013 May;144(5);903–911. PMID: 23357715 View Abstract
- Biesiekierski JR, Newnham ED, Irving PM, Barrett JS, Haines M, Doecke JD, Shepherd SJ, Muir JG, Gibson PR: Gluten causes gastrointestinal symptoms in subjects without celiac disease: a double-blind randomized placebo-controlled trial. Am J Gastroenterol 2011 Mar;106(3):508-14; quiz 515. PMID: 21224837 View Abstract
- Biesiekierski JR, Peters SL, Newnham ED, Rosella O, Muir JG, Gibson PR: No effects of gluten in patients with self-reported non-celiac gluten sensitivity after dietary reduction of fermentable, poorly absorbed, short-chain carbohydrates. Gastroenterology 2013Aug;145:320-8.e1-3. PMID: 23648697 View Abstract
- Junker Y, Zeissig S, Kim S, Barisani D, Wieser H, Leffler DA, Zevallos V, Libermann TA, Dillon S, Freitag TL, Kelly CP, Schuppan D: Wheat amylase trypsin inhibitors drive intestinal inflammation via activation of toll-like receptor 4. J Exp Med 2012 Dec;209(13):2395-408. PMID: 23209313 View Abstract
- www.allergysf.com/allergy-education/big-8-top-food-allergens/
- http://www.mayoclinic.org/diseases-conditions/wheat-allergy/basics/definition/con-20031834
- Whitaker, J.K.H., West, J., Holmes, G.K.T. & Logan, R.F.A. (2009) Patient perceptions of the burden of celiac disease and its treatment in the UK. Aliment. Pharmacol. Ther. 29, 1131–1136. PMID: 19245681 View Abstract
- Zarkadas M, Dubois S, MacIsaac K, Cantin I, Rashid M, Roberts KC, La Vieille S, Godefroy S, Pulido OM. Living with coeliac disease and a gluten-free diet: a Canadian perspective.J Hum Nutr Diet. 2013 Feb;26(1):10-23PMID: 23157646 View Abstract
- Roos S, Kärner A, Hallert C. Gastrointestinal symptoms and well-being of adults living on a gluten-free diet: a case for nursing in celiac disease. Gastroenterol Nurs. 2009 May-Jun;32(3):196-201. PMID: 19506436 View Abstract
- Rubio-Tapia A, Rahim MW, See JA, Lahr BD, Wu TT, Murray JA. Mucosal recovery and mortality in adults with celiac disease after treatment with a gluten-free diet. Am J Gastroenterol. 2010 Jun;105(6 Jan):1412-20. PMID: 20145607 View Abstract
- Hollon JR, Cureton PA, Martin ML, Puppa EL, Fasano A. Trace gluten contamination may play a role in mucosal and clinical recovery in a subgroup of diet-adherent non-responsive celiac disease patients BMC Gastroenterol. 2013; 13: 40. PMID: 359883967
- http://glutenfreepassport.com/living-gluten-allergy-free/food-product-labeling/
- Catassi C, Fabiani E, Iacono G, et al. A prospective, double-blind, placebo-controlled trial to establish a safe gluten threshold for patients with celiac disease. Am J Clin Nutr 2007; 85(1): 160-6. PMID: 17209192 View Abstract
- Catassi C, Rossini M, Ratsch IM, et al. Dose dependent effects of protracted ingestion of small amounts of gliadin in coeliac disease children: a clinical and jejunal morphometric study. Gut 1993 Nov; 34(11): 1515-9. PMID: 8244135 View Abstract
- http://www.glutenfreedietitian.com/how-much-gluten-is-20-parts-per-million/
- Szaflarska-Popławska A. Non-dietary methods in the treatment of celiac disease Prz Gastroenterol. 2015;10(1):12-7. PMID: 25960809 View Abstract
- Leonard MM, Vasagar B. US perspective on gluten-related diseases. Clin Exp Gastroenterol. 2014 Jan 24;7:25-37. PMID: 24493932 View Abstract
- Prasad KK, Thapa BR, Nain CK, Sharma AK, Singh K. Brush border enzyme activities in relation to histological lesion in pediatric celiac disease. J Gastroenterol Hepatol. 2008 Aug;23(8 Pt 2):e348-52. PMID: 18070009 View Abstract
- Personal conversation, September, 201575.
- Szaflarska-Popławska A Non-dietary methods in the treatment of celiac disease. Prz Gastroenterol. 2015;10(1):12-7. PMID: 25960809 View Abstract
- Detel D, Persić M, Varljen J. Serum and intestinal dipeptidyl peptidase IV (DPP IV/CD26) activity in children with celiac disease. J Pediatr Gastroenterol Nutr. 2007 Jul;45(1):65-70. PMID: 17592366 View Abstract
- Thoma R, et al. Structural basis of proline-specific exopeptidase activity as observed in human dipeptidyl peptidase-IV. Structure. 2003 Aug;11(8):947-59. PMID: 12906826 View Abstract
- Janssen G, et al. Ineffective degradation of immunogenic gluten epitopes by currently available digestive enzyme supplements. PLoS One. 2015 Jun 1;10(6):e0128065. PMID: 26030273 View Abstract
- Stoven S, Murray JA, Marietta E. Celiac Disease: Advances in Treatment via Gluten Modification. Clinical Gastroenterology & Hepatology. 2012 Aug;10(8):859–62. PMID: 22728383 View Abstract
- Mitea C, et al. Efficient degradation of gluten by a prolyl endoprotease in a gastrointestinal model: implications for coeliac disease. Gut. 2008 Jan;57(1):25-32. PMID: 17494108 View Abstract
- Montserrat V, Bruins MJ, Edens L, Koning F. Influence of dietary components on Aspergillus niger prolyl endoprotease mediated gluten degradation. Food Chem. 2015 May 1;174:440-5. PMID: 25529703 View Abstract
- Ehren J, Morón B, Martin E, Bethune MT, Gray GM, Khosla C. A food-grade enzyme preparation with modest gluten detoxification properties. PLoS One. 2009 Jul 21;4(7) .PMID: 19621078 View Abstract
- Salden B, Monserrat V, Troost V, Bruins MJ, Edens L, Bartholom R, Haenen B, Winkens B, Koning F, Masclee AA. Randomised clinical study: Aspergillus niger-derived enzyme digests gluten in the stomach of healthy volunteers. Aliment Pharmacol Ther 2015 Aug; 42: 273–285. PMID: 26040627 View Abstract
- Hudson M, King C. Unpublished clinical trial, Glutalytic Clinical Trial for Normal Consumption of Gluten Containing Foods, Department of Biology, Kennesaw State University, Kennesaw, GA
- Volta U, Tovoli F, Cicola R, Parisi C, Fabbri A, Piscaglia M, Fiorini E, Caio G Serological tests in gluten sensitivity (nonceliac gluten intolerance). J Clin Gastroenterol. 2012 Sep;46(8):680-5. PMID: 22138844 View Abstract
- Tolerance and Efficacy of Glutalytic: A Randomized, Double-Blind, Placebo Controlled Study. publication forthcoming in 2016.
- Personal conversation, April, 2015.