 Obesity: A consequence of adverse inflammation & microbial disruption?
Obesity: A consequence of adverse inflammation & microbial disruption?
By Michael Ash BSc(Hons) DO, ND, FDipION
Published in CAM 2005
Overweight and obesity are serious, chronic medical condition associated with a wide range of debilitating and life threatening and economically burdensome conditions. The recent and extensive increases in obesity among Europeans are eroding many recent health gains.
Paradoxically the economically wealthier communities of the world continue to over consume food and food products, whilst other nation communities still suffer from food deprivation and starvation, due in the main to drought, floods, ‘acts of God’, corruption and conflict. Approximately 9.5% of the global burden of disease is currently attributable to being underweight,[1] whilst there are now hundreds of millions of people (>500) in developed and developing countries that are overweight or obese. This condition of excessive weight is now so common that it is rapidly replacing malnutrition and infectious diseases as the most significant cause of ill health[2]. An escalating global epidemic of overweight and obesity – “globesity” – is taking over many parts of the world.
This growing epidemic has manifested as huge increases in body mass index (BMI) in most, if not all, countries, and is due to a combination of differing influences, the most commonly perceived ones being “over nourishment” and “under activity”.
Obesity is a multifactorial condition understood to be a result of the interplay between internal and external environments, psychosocial influences, dietetic choices, immune activation and genetic tendencies. The rate of its development in the latter half of the 20th century and its apparent ongoing speed of expansion in the 21st century requires innovative and effective intervention.
Compelling evidence identifying the rapid rate of change in obesity prevalence comes from various countries, including North America, the United Kingdom and Australia. In the United States, the Centers for Disease Control and Prevention have reported 60% of the population have a BMI > 25 kg/m2, and 27% are obese.[3] This reflects a substantive 61% increase in obesity since 1991, a period covering only 8 years in this study.[4]
Similar trends have been observed in Canada, where the prevalence of obesity increased from 10% in 1970–72 to 15% in 1998.[5] The UK has not escaped the expansion of adiposity with the prevalence of obesity rising among men from 13% in 1993 to 21% in 2000, and among women from 16% to 21%.[6]
Innovative interventions, manageable and sustainable ongoing strategies including selective macro nutrient intake and increased physical activity are required to halt and reverse this condition. The individual with a normal BMI is now in the minority and is likely to become a rarity in the future without substantive nationwide change.
In the search for resolution of obesity there have been extensive proposals to explain the apparent insatiable demand for the growth our bodies’ adipocytes have achieved. There are many sound and tested concepts and beliefs, but the fact remains we are losing the battle.
Our genome it seems is hard wired for storage; the era of food abundance in evolutionary terms has covered only a short time span in comparison to the extended period of inadequate food availability our species has endured. We have yet to persuade our genetic code to fail to prepare our adipocytes for a prolonged period of food deprivation. Consequently the central DNA make up involved in energy storage appears to have been pushed into a state of over expression through it’s constant exposure to gene modifying factors, including food choice, rate and type of consumption, persistent inflammation, aberrant immune activity and intestinal microbial change[7].
The research exploring the evolutionary theories has focused on the potential survival advantages of “thrifty” genes and has concluded they are now maladaptive or over expressed. Other important and modifiable factors have been exposed through many mechanistic studies which have revealed numerous fat-derived molecules and a link to chemicals associated with inflammation that, together, underlie the obesity-diabetes connection and represent for clinicians, prospective targets for therapeutic intervention.
So next time you are considering how to manage the weight loss resistant patient or even your own difficulties with weight, it may be clinically advantageous to think outside the traditional models.
One component of our being covers the combination of the above; the cross talk between our genetic make up and our co evolutionary partners, our microbiota, appears to be an area of profound opportunity for genetic nutritioneering[8]. A second and associated component, the immune regulatory disturbances so noted at the mucosal tissues, when innate immunity loses its ability to manage adaptive immune responses in a reasonable manner is also a key factor. The common site for these mixed impacts is found in the Common Mucosal Immune System (CMIS) and most specifically in the gastro intestinal tract (GIT) and its composite bacterial bed fellows.
Obesity is a disease of inflammation and disturbed microbial balance
The bulk of research examining the increased burden of inflammation within the obese patient has to date considered the source of excess cytokines and adipokines as the sole responsibility of the adipocytes. Current opinion is now evolving from single source production to a multi-factorial origination with the adaptive and innate immune systems as significant contributors[9]. This evolving comprehension is now expanding the role of mucosal immunity as an important component in systemic pathologies including insulin resistance, syndrome X and obesity. Disruption of the mucosal anti inflammatory function through down regulation of secretory immunoglobulin A (SIgA) will lead to increased production of inflammatory cytokines[10].
Although once considered a metabolically inactive fuel depot, adipose tissue is now recognised to be an endocrine organ communicating with the brain and peripheral tissues through the secretion of various hormones regulating appetite and metabolism. By virtue of its mass it is the largest endocrine organ in the body[11]. These functions are modulated and determined by the location of the adipose tissue (visceral versus subcutaneous)[12] by the mass of the average adipocyte present in the tissue[13]and by the adipocytes metabolism of glucose[14] and corticosteroids[15].
One such adipokine (fat derived cytokine) is adiponectin a hormone with antidiabetic properties. The ability of adiponectin to increase insulin sensitivity in conjunction with its anti-inflammatory and anti-atherogenic properties has made this novel adipocytokine a promising therapeutic tool[16]. Adiponectin one of the beneficial adipokines induces increased sensitivity to insulin but is down regulated in the face of inflammation and reactive oxygen species (ROS)[17].
Modifying the production of this protein as well as minimising those less helpful agents constitutes a potential target for therapies aimed at uncoupling insulin resistance from obesity.
The plant protein osmotin found in fruits, nuts and vegetables and used by them as part of their innate defence has the effect of mimicking adiponectin in humans by binding to its receptor sites.[18] Osmotin is a stable protein resistant to heat, acidity and enzymes, meaning it could circulate through the body without being broken down by digestion. Whether osmotin plays any role in the health benefits attributed to diets high in fruits and vegetables is a tantalising possibility, certainly the maxim of eating more fruits and vegetables has many effector events within the human
Adiponectin and osmotin jump-start a process called AMP kinase phosphorylation, which increases sugar and fat use by muscle cells. By binding to the adiponectin receptor, osmotin, like adiponectin, can control the energy status of muscle cells.[19]
TNFa and IL6 Cytokines of immunity and obesity
Cytokines are intercellular signalling polypeptides produced by activated cells. Most cytokines have multiple sources, multiple targets, and multiple functions. Typical properties of cytokines in these networks are pleiotropy, redundancy, synergistic activity and antagonistic effects upon each other[20]. Inflammation-associated cytokines include interleukin-6, interleukin-1ß, tumor necrosis factor, interferon-, transforming growth factor ß,[21] and possibly interleukin-8.[22]
They are produced by a variety of cell types, but the most important sources are macrophages and monocytes at inflammatory sites.[23].The cytokines TNFa and IL6 are extensively produced by macrophages as well as by adipocytes; they act directly on inflammatory cells and also contribute indirectly to inflammation by acting on the liver to produce acute phase proteins. These cytokines also induce a suppressor of cytokine signaling-3 (SOCS-3), an intracellular signalling molecule that impairs the signalling of both leptin (a cytokine that acts as a moderator of food intake, and acts as a neuro endocrine hormone, blunted in the face of up regulated inflammation) and insulin. SOCS-3 levels are elevated in obesity and thus may represent a final common pathway of obesity-associated resistance to the actions of both leptin and insulin[24].
TNFa has been strongly implicated as a developmental molecule in insulin resistance and obesity[25]. It appears to be ‘over expressed’ in adipocytes of obese individuals and reduces cell receptor sensitivity to insulin as well as influencing transcription of specific adipocyte genes. This cytokine may be induced locally within the adipocyte for a variety of hormone regulatory actions and triglyceride induction, but the single largest producer of TNFa is the adaptive aspect of the cellular immune system, the largest mass of immune tissues present within the mucosal barriers of the gastrointestinal tract.
TNFa is an essential component of mucosal immunity where it has a dual role defending us and yet if allowed to be produced unchecked it has the ability to create intense health problems in the host if left unchecked, including at the worst outcome a loss of life through sepsis[26].
TNFa, it seems is more than an immune defence cytokine. Its impact is felt across many tissues and in particular it has powerful effects on insulin resistance in obesity through its effects on several important sites of insulin action[27].
Increased production of these cytokines is found in the presence of disrupted mucosal immunity and exposure to microbial or antigenic provocateurs that interfere with the normal state of immune tolerance[28]. A failure to maintain correct mucosal tolerance through the loss of innate immune system cross talk will always result in the increased production of inflammatory cytokines. Maintaining a healthy and functional micro ecology and adequate production of the immunoglobulin Secretory IgA is essential to achieve this state.
NFkB The inflammation amplifier
The gene transcription factors IKKb and NFkB are molecular mediators of insulin resistance and also represent a target for therapeutic intervention[29]. The NFkB cascade is a major pathway for the amplification of inflammatory processes.[30] NFkB is a ubiquitous nuclear transcription factor that promotes the activation of genes that encode for inflammatory mediators and enzymes, NFkB can be thought of as the major intracellular “amplifier” which ultimately increases the production of the direct mediators of inflammation such as cytokines, prostaglandins, leukotrienes, nitric oxide and other reactive oxygen species (ROS).
Understanding this concept provides clinicians with a unique approach to their adipose abundant patients, for whilst inflammation is well understood to be a predictive risk indicator for declining insulin receptor sensitivity, it must also be considered as a therapeutic ‘foot in the door’ as modifying production of inflammatory cytokines appears to improve the ability to switch genetic expression off or at least down regulate its willingness to store fat.
Intestinal macrophages normally eat without getting upset
Our intestinal macrophages, derived from blood monocytes, are thought to conduct control over mucosal inflammatory responses. However, Lesley Smythies and colleagues have now demonstrated that resident intestinal macrophages lack many innate response receptors and do not actually produce the proinflammatory cytokines such as IL1, IL6, IL8, and TNF during normal states of tolerance, although the cells retain certain immune capacity through phagocytic and bacteriocidal activity 21.
The inflammatory ‘anergy’ of intestinal macrophages, reflected in the cells’ reduced expression of innate response receptors and their down regulated production of cytokines, indicates a potential mechanism for the absence of inflammation in normal intestinal mucosa despite the close proximity of immunostimulatory bacteria and LPS. In effect, they are able to contribute to the mediation of ‘mucosal tolerance’ through appropriate anergic response activity.
The resultant inflammatory anergy serves to limit mucosal inflammation in normal intestinal mucosa, whereas the loss or dysregulation of macrophage inflammatory anergy may promote the inflammation associated with inflammatory bowel disease[31]. Maintaining mucosal tolerance is one of the key roles of SIgA and bacterial commensals and is also dependant on food choice, volume of antigen and microbial exposure.
Oxidative Stress
Oxidative stress may be also be induced by obesity and increased oxidative stress in accumulated fat is at least in part responsible for the dysregulation of adipocytokines and the eventual development of metabolic syndrome with its associated hyperglycaemia, dyslipidaemia, hypertension and atherosclerosis. Obesity is, of course closely associated with metabolic syndrome[32].
Fat accumulation is closely correlated with markers of systemic oxidative stress, which in turn correlates with body mass index (BMI)[33] The insulin sensitising adipocytokines in plasma have an inverse correlation with systemic oxidative stress.
The close relationship between inflammation and diabetes is supported by the observation that stimulation of the innate immune response (by bacterial endotoxin during sepsis, for example)[34] results in insulin resistance that contributes to the high mortality of critical illness[35] . The interaction between inflammation and insulin signalling is also suggested by the ability of aspirin to improve insulin resistance, in part by preventing the antagonistic effects of fatty acids and cytokines[36].
Why is Obesity is a disease of inflammation
Why is obesity an inflammatory state and why does inflammation cause diabetes? The search for answers to these questions takes us again to evolutionary considerations. Perhaps the response to infection is more effective when glucose is shunted from muscle to the inflammatory cells involved in the immune response and tissue repair.
A potentially unifying view is that the body’s ability to survive major stress, including infection and starvation, is enhanced by peripheral insulin resistance that preserves the brain’s glucose supply[37]. This hypothesis might explain why cortisol, the major stress hormone, causes insulin resistance and stimulates the innate immune response12 , even though chronic cortisol exposure is anti-inflammatory because of down-modulation of the acquired immune response.
The stress connection may extend to individual cells, as it has recently been shown that intracellular stress induces insulin resistance in a manner that is exacerbated by obesity, potentially through adipocyte-secreted factors[38]. Moreover, chronic metabolic stress impairs the ability of pancreatic beta cells to secrete sufficient insulin to overcome insulin resistance, which is a hallmark of type 2 diabetes[39].
Our body produces a wide range of cytokines, proteins and other communication molecules. The gastro intestinal tract (GIT) is responsible for the origination of many of these. The interplay between those induced or stimulated in the GIT and elsewhere makes for clinically interesting connections and functional interaction. Certain of these cytokines are now well known to have an impact on weight and metabolic syndrome, others are less well understood.
The interconnectiveness of the mucosal tissues through their many layered communication networks including microvasculature, enteric and central nervous system and immune cells establishes them as a prime site for investigation and development of clinical interventions for their modification.
Advanced Glycosolation End Products AGE’s
Inflammatory chemicals are also promoted by dietary glycotoxins[40]. Diet is a major source of the proinflammatory chemicals AGE’s (heat generated advanced glycation end products) These AGE’s are created in common foods during spontaneous reactions between reducing sugars and proteins or lipids[41].
The inclusion of supplemental antioxidants adds further control to the production of proinflammatory cytokines indicating that dietetic modulation and enhancement of antioxidant status at the GIT produces diminished circulating inflammatory markersError! Bookmark not defined.. Inappropriate immune and bacterial associated immune regulation induces increased ROS[42] and contributes to amplified problems associated with diabetes and may well increase the development of obesity.
Increased oxidative stress can now be linked to food choice, volume and composition, in addition immune activation at the tissue sites these macro molecules are meeting will contribute to upregulated reactive oxidative species production and other chemicals aimed at achieving immune tolerance.42
Bugs Make YOU Fat
Humans and other mammals are colonised by a vast, complex, and dynamic consortium of micro-organisms. One of the evolutionary driving forces for maintaining this metabolically active microbial biomass is to aid in the salvage of carbohydrates that would otherwise be indigestible.
This complex dynamic collection predominately found in the GIT means we are more prokaryotic than eukaryotic as estimates define us as being 90% microbial cell and 10% human cell.[43] This collection of organisms may as a result be perceived of as being a ‘metabolically’ active organ, playing a critical role in nutrition and energy expenditure and storage.[44]
This intestinal eco system is constantly shaped by interactions between its microbes (both intra and inter species communications), epithelium, mucosal immune system, microvasculature and enteric nervous system.[45] Initially sterile the human gut goes through dramatic changes in it gut microflora until it reaches its ‘climax community’ which in turn remains highly modifiable by environmental and host gene factors.[46]
The commensal (‘at the table together’) is particularly apropos with the intestinal microflora as both depend on the dietetic intake from the host. One might think that both host and microflora would now engage in competition for the same nutrients. However, conventionally raised animals with an intact biomass require 30% less caloric intake to maintain their body weight than their germ free counterparts, indicating that the microbiota is involved in nutrient optimisation.[47]
This concept exposes the question ‘is the composition of microflora a risk factor for obesity?’ The implication being that the caloric value of a meal should not be seen as a fixed entity, but rather as a value influenced by the consumers’ intestinal microbiota. In order to fully define our metabolic potential we would have to include that of our microbial colleagues, as these master ‘physiologic chemists’ have over our evolutionary history developed complex chemical strategies for regulating nutrient processes to the benefit of them and the host. After all we originated from single cell evolution and the earth has been populated with bacteria for over 2.5 billion years[48].
One of these ancient host signalling pathways appears to have a potential implication for fat storage and obesity. Research conducted by Washington University Medical Dept led by Prof Gordon has demonstrated that inclusion of a dietetically similar mice chow diet to germ free mice against conventionally microbiota populated mouse revealed a huge 42% increase in total body fat in the microbially populated mice despite a 29% reduction in calorific intake.
When microbiota harvested from conventionalised mice was then added to 8-10 week old germ free mice the effect was again dramatic with a 61% increase in body fat against their GF counterparts despite 27% decreased chow consumption. Both sexes were affected with female mice showing a higher deposition.
In addition there was increased insulin resistance and increased triglycerides, as well as clear evidence of increased monosaccharide uptake. The transport of this nutrient to the liver is assisted by the microbiota induced increase in capillary density.[49] Once there the liver can either increase inefficient metabolism or export for peripheral storage as fat. The increase in fat storage resulted in enlarged adipocytes, and circulating fatty acids which in turn diminish availability of leptin for the brain and reducing glucose uptake by cells.
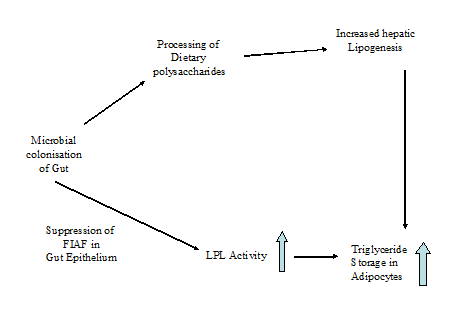
Lipoprotein Lipase (LPL) is a key regulator of fatty acid release from triglyceride-rich lipoproteins in muscle, heart, and fat[50] increased adipocyte LPL activity leads to increased cellular uptake of fatty acids and adipocyte triglyceride accumulation. The increase fat storage is mediated by a protein like chemical called FIAF (Fasting Induced Adipocyte Factor). FIAF appears to have been preserved over the course of vertebrate evolution[51]. FIAF is one of many host factors influenced by gut microbes. There are 10-100 trillion microbes co-existing in the adult human gut, and this vast community of microbes has evolved ways of manipulating our biology to benefit them and us.
In fact, the gut microbiota may be considered as one of the body’s organs, one exquisitely attuned to our physiological needs.
“Over their millions of years of co-evolution with us, microbes have learned to manipulate networks of human genes,”. “By defining these networks, we can learn ‘new ways’ to promote health.”
The ability to store foods would have been advantageous to ancient humans who had variable access to food, in contemporary society, the ready access means this feature becomes a disadvantage. In the face of specific bacterial colonisation or overgrowth via provocational exposure to preferred food substrates FIAF is suppressed leading to fat storage.
Lower colonic bacteria appear to be the controlling organisms, and the rate appears dependant on population mix and exposure to polysaccharides[52]. The researchers discovered that the lean, bacteria-free rodents had more of the enzyme called fasting-induced adipocyte factor (FIAF) produced in the gut lining, liver, and fat cells. It lowers the amount of fat in these cells, but when Bacteroides is hard at work in the gut, FIAF is suppressed and absent, and fat cells load up increasing adiposity. FIAF represents a novel endocrine signal involved in the regulation of metabolism and is found in the plasma in higher levels during fasting[53].
Bacteroides thetaiotaomicron is the species currently associated with the manipulation of FIAF its genome is one of the largest examined, and it is dedicated to breaking down complex carbohydrates that enzymes in the human gut cannot otherwise process. The complex carbohydrates are polysaccharides, common to foods such as vegetables and other plant materials. Our bodies should consume 15 to 20 percent of daily caloric intake this way, but many dietary choices will adversely affect this ratio. The bacteria also exhibit an elaborate environment-sensing system to help them thrive in the highly competitive community within the human digestive system.
In order to be successful in the gut where there are literally trillions of bacteria competing for the nutrients their human host eats, the successful microbe must develop a system to determine what nutrients to go after and a strategy to capture them. B. thetaiotaomicron has evolved to do this with great facility. It has developed a unique ability to grab polysaccharides in the GIT and then release specific enzymes to harvest these nutrients as they enter its mucosal niche. The results benefit the host and the organisms. It is this competence at managing polysaccharides that appears to add complications when suppression of FIAF is also associated with this species.
These findings imply that microbial suppression of intestinal FIAF promotes adiposity, and suggests that increasing FIAF expression and/or activity may promote leanness. It is reasonable to speculate that changes in microbial ecology prompted by Western diets, and medication use and/or differences in microbial ecology between individuals living in these societies, may function as an “environmental” factor that affects predisposition toward energy storage and obesity.
Case History
Patient J, a 57 year old obese self employed male, with insulin resistance, hypertension, hypercholestralaemic, raised triglycerides and with mild depression.
Had followed a dietetic and lifestyle programme using macro food adjustments and exercise regimes as well as blood sugar and adrenal nutritional support. Successfully reduced the weight from 24 stone to 21 stone but had been unsuccessful in reducing further his weight or markers of metabolic syndrome.
In an attempt to see if competitively inhibiting Thetaiotaomicron and managing the mucosal immune system would have any effect he was given a diet with slightly increased protein intake, 750mg of Saccharomyces Boulardii to raise SigA and 90 billion Lactic acid bacteria (Culturelle) and 60 billion Bacteroides without FOS (BifidoBactT) per day.
In 30 days he lost 8lb in weight, had a 25% reduction in BP, and noticed a significant change in mood and energy. Three months on he has lost 1 stone, maintained his BP reduction and reduced medication, mood has remained buoyant and energy is high, insulin resistance has declined and triglycerides has also fallen.
This single case illustrates that there may be a route to manipulate entrenched weight and associated dysfunction via the master chemists living inside our body and assisting them in maintaining an effective level of mucosal tolerance.
References
[1] World Health Organization. The World Health Report 2002. Geneva: WHO, 2002.
[2] World Health Organization. Obesity: preventing and managing the global epidemic. Report of a WHO consultation. World Health Organ Tech Rep Ser 2000; 894: 1-253.
[3] Centres for Disease Control and Prevention. Prevalence of overweight and obesity among adults: United States. Atlanta: National Centre for Health Statistics, 1999.
[4] Mokdad AH, Bowman BA, Ford ES, et al. The continuing epidemics of obesity and diabetes in the United States. JAMA 2001; 286: 1195-2000.
[5] Katzmarzyk PK. The Canadian obesity epidemic, an historical perspective. Obes Res 2002; 10: 666-674
[6] Joint Health Survey Unit. Health surveys for England, 2000. London: National Centre of Social Research and the Department of Epidemiology and Public Health, University College, London, 2001.
[7] Fredrik Backhed, Hao Ding, Ting Wang~, Lora V. Hooper, Gou Young Koh, Andras Nagy Clay F. Semenkovich, and Jeffrey I. Gordon. The gut microbiota as an environmental factor that regulates fat storage. 15718–15723 ~ PNAS ~ November 2, 2004 ~ vol. 101 ~ no. 44
[8] Kaput J, Rodriguez RL. Nutritional genomics: the next frontier in the postgenomic era. Physiol Genomics. 2004 Jan 15;16(2):166-77
[9] Plevy S. The immunology of inflammatory bowel disease. Gastroenterol Clin North Am. 2002 Mar;31(1):77-92
[10] Fernandez MI, Pedron T, Tournebize R, Olivo-Marin JC, Sansonetti PJ, Phalipon A. Anti-inflammatory role for intracellular dimeric immunoglobulin a by neutralization of lipopolysaccharide in epithelial cells. Immunity. 2003 Jun;18(6):739-49
[11] E. E. Kershaw, J. S. Flier, J. Adipose Tissue as an Endocrine Organ. Clin. Endocrinol. Metab. 89, 2548 (2004)
[12] M. Das, I. Gabriely, N. Barzilai, Caloric restriction, body fat and ageing in experimental models Obes. Rev. 5, 13 (2004).
[13] . C. Weyer et al., Subcutaneous abdominal adipocyte size, a predictor of type 2 diabetes, is linked to chromosome 1q21–q23 and is associated with a common polymorphism in LMNA in Pima Indians. Mol. Genet. Metab. 72, 231 (2001)
[14] E. D. Abel et al., Adipose-selective targeting of the GLUT4 gene impairs insulin action in muscle and liver Nature 409, 729 (2001)
[15] H. Masuzaki et al., A transgenic model of visceral obesity and the metabolic syndrome.
Science. 2001 Dec 7;294(5549):2166-70Science 294, 2166 (2001
[16] Nedvidkova J, Smitka K, Kopsky V, Hainer V. Adiponectin, an adipocyte-derived protein. Physiol Res. 2004 Nov 15
[17] Yamauchi, T., et al 2001. The fat derived hormone adiponectin reverses insulin resistance associated with both lipoatrophy and obesity.: Nat. Med 7:941-946
[18] Narasimhan ML, Coca MA, Jin J, Yamauchi T, Ito Y, Kadowaki T, Kim KK, Pardo JM, Damsz B, Hasegawa PM, Yun DJ, Bressan RA. Osmotin Is a Homolog of Mammalian Adiponectin and Controls Apoptosis in Yeast through a Homolog of Mammalian Adiponectin Receptor. Mol Cell. 2005 Jan 21;17(2):171-80.
[19] Narasimhan ML, Coca MA, Jin J, Yamauchi T, Ito Y, Kadowaki T, Kim KK, Pardo JM, Damsz B, Hasegawa PM, Yun DJ, Bressan RA. Osmotin Is a Homolog of Mammalian Adiponectin and Controls Apoptosis in Yeast through a Homolog of Mammalian Adiponectin Receptor. Mol Cell. 2005 Jan 21;17(2):171-80.
[20] Townsend MJ, McKenzie AN. Unravelling the net ? cytokines and diseases. J Cell Sci. 2000 Oct;113 (Pt 20):3549-50
[21] Kushner I. Regulation of the acute phase response by cytokines. Perspect Biol Med 1993;36:611-622
[22]Wigmore SJ, Fearon KCH, Maingay JP, Lai PBS, Ross JA. Interleukin-8 can mediate acute-phase protein production by isolated human hepatocytes. Am J Physiol 1997;273:E720-E726
[23] M. W. Rajala et al., Regulation of Resistin Expression and Circulating Levels in Obesity, Diabetes, and Fasting Diabetes 53, 1671 (2004)
[24] H. Shi, I. Tzameli, C. Bjorbaek, J. S. Flier, J. Biol. Chem. 279, 34733 (2004)
[25] Um JY, Kang BK, Lee SH, Shin JY, Hong SH, Kim HM. Polymorphism of the tumor necrosis factor alpha gene and waist-hip ratio in obese Korean women. Mol Cells. 2004 Dec 31;18(3):340-5
[26] Andersson J. The inflammatory reflex – Introduction. J Intern Med. 2005 Feb;257(2):122-5
[27] Teoman Uysal K, Weisbrock S, et al; Protection from obesity induced insulin resistance in mice lacking TNFa function: Nature 389, 610-614 9th Oct 1997; Doi: 10.1038/39335
[28] Sitaraman SV, Merlin D, Wang L, Wong M, Gewirtz AT, Si-Tahar M, Madara JL. Neutrophil-epithelial crosstalk at the intestinal lumenal surface mediated by reciprocal secretion of adenosine and IL-6. J Clin Invest. 2001 Apr;107(7):861-9
[29] Shoelson S, Lee J, Yuan M; Inflammation and the IKKb/IkB/NFkB axis in obesity and diet-induced insulin resistance. Int J Obesity Vol 27, suppl 3, s49-s52 Dec 2003. DOI: 10.1038/sj.ijo.0802501
[30] D’Acquisto F, May MJ, Ghosh S. Inhibition of Nuclear Factor Kappa B (NF-B):: An Emerging Theme in Anti-Inflammatory Therapies. Mol Interv. 2002 Feb;2(1):22-35 Available athttp://molinterv.aspetjournals.org/cgi/reprint/2/1/22 on November 9, 2004
[31] Lesley E. Smythies, Marty Sellers, Ronald H. Clements, et al Human intestinal macrophages display profound inflammatory anergy despite avid phagocytic and bacteriocidal activity J. Clin. Invest. 115:66-75 (2005).
[32] Ford E S., Giles W H., Dietz W H., 2002 Prevalence of the metabolic syndrome among US adults: Findings from The Third National Health and Nutritional Examination Survey JAMA. 287:356-359
[33] Keaney JF., et al. 2003. Obesity and systemic oxidative stress: clinical correlations of oxidative stress in the Framingham Study. Arterioscler. Thromb. Vasc. Biol. 23:434-439
[34] A. O. Agwunobi, C. Reid, P. Maycock, R. A. Little, G. L. Carlson, Insulin Resistance and Substrate Utilization in Human Endotoxemia J. Clin. Endocrinol. Metab. 85, 3770 (2000)
[35] G. Van den Berghe, How does blood glucose control with insulin save lives in intensive care? J. Clin. Invest. 114, 1187 (2004)
[36]M. Yuan et al., Reversal of Obesity- and Diet-Induced Insulin Resistance with Salicylates or Targeted Disruption of Ikk beta Science 293, 1673 (2001).
[37] P. H. Black, The inflammatory response is an integral part of the stress response: Implications for atherosclerosis, insulin resistance, type II diabetes and metabolic syndrome X. Brain Behav. Immun. 17, 350 (2003)
[38] U. Ozcan et al., Endoplasmic Reticulum Stress Links Obesity, Insulin Action, and Type 2 Diabetes Science 306, 457 (2004)
[39] C. J. Rhodes, Type 2 Diabetes-a Matter of ß-Cell Life and Death? Science, 307, 380
[40] Monnier VM, Obrenovich ME. Wake up and smell the maillard reaction. Sci Aging Knowledge Environ. 2002 Dec 18;2002(50):pe21
[41] Baynes J.W & Thorpe, S.R 2000 Free Rad Biol. Med. 28, 1708-1716
[42] Acheson DW, Luccioli S. Microbial-gut interactions in health and disease. Mucosal immune responses. Best Pract Res Clin Gastroenterol. 2004 Apr;18(2):387-404
[43] Savage DC. 1977. Microbial ecology of the gastrointestinal tract. Annu. Rev. Microbiol. 31:107–33
[44] Savage DC. 1986. Gastrointestinal microflora in mammalian nutrition. Annu. Rev. Nutr. 6:155–78
[45] Gordon JI, Hooper LV, McNevin MS, Wong M, Bry L. 1997. Epithelial cell growth and differentiation. III. Promoting diversity in the intestine: conversations between the microflora, epithelium, and diffuse GALT. Am. J. Physiol. Gastrointest. Liver Physiol. 273:G565–G70
[46] Zoetendal EG, Akkermans AD, Akkermans-van Vliet WM, de Visser J, de Vos WM. 2001. The host genotype affects the bacterial community in the human gastrointestinal tract. Microb. Ecol. Health Dis. 13:129–34
[47] Wostmann BS, Larkin C, Moriarty A, Bruckner-Kardoss E. 1983. Dietary intake, energy metabolism, and excretory losses of adult male germfree Wistar rats. Lab Anim. Sci. 33:46–50
[48] J. J. Brocks, G. A. Logan, R. Buick, R. E. Summons, Science 285, 1033 (1999).
[49] Stappenbeck, T. S., Hooper, L. V. & Gordon, J. I. (2002) Proc. Natl. Acad. Sci. USA 99, 15451–15455.
[50] Preiss-Landl K, Zimmermann R, Hammerle G, Zechner R. Lipoprotein lipase: the regulation of tissue specific expression and its role in lipid and energy metabolism. Curr Opin Lipidol. 2002 Oct;13(5):471-81
[51] Rawls, J. F., Samuel, B. S. & Gordon, J. I. (2004) Proc. Natl. Acad. Sci. USA 101, 4596–4601.
[52] Fredrik Backhed, Hao Ding, Ting Wang~, Lora V. Hooper, Gou Young Koh, Andras Nagy Clay F. Semenkovich, and Jeffrey I. Gordon. The gut microbiota as an environmental factor that regulates fat storage. 15718–15723 ~ PNAS ~ November 2, 2004 ~ vol. 101 ~ no. 44
[53] Kersten S, Mandard S, Tan NS, Escher P, Metzger D, Chambon P, Gonzalez FJ, Desvergne B, Wahli W. Characterization of the fasting-induced adipose factor FIAF, a novel peroxisome proliferator-activated receptor target gene. J Biol Chem. 2000 Sep 15;275(37):28488-93






