 How Oxidative Stress and the Hypothalamic-Pituitary-Adrenal Axis May Play a Role by Dr Carrie Decker ND
How Oxidative Stress and the Hypothalamic-Pituitary-Adrenal Axis May Play a Role by Dr Carrie Decker ND
Addiction: A Stressed, Nutritionally Depleted System
It likely is not a stretch to envision the stressors that the entire human body and mind must endure during battles with addictive substances and/or behaviours. Be it a drug like heroin or cocaine, or a behaviour such as gambling, the addiction takes front and centre. Biological necessities such as sleep and eating recede into the background, predisposing to nutritional deficiencies. Subclinical or even gross malnutrition puts every cell under a state of stress as the body tries to maintain normal function in a state of deficiency. Although nutritional status varies considerably among individuals with substance use disorders and is impacted by the substance of choice, use patterns,[1] and socioeconomic factors,[2] certain facts are worthy of note.
When the substance of choice is alcohol or a drug, the burden on the liver and the kidneys to detoxify these toxins easily multiplies by many factors the encumbrances of nutritional deficiencies.[3] Excessive alcohol intake alters lipid, glucose, and protein metabolism: it inhibits lipolysis, promotes glucose intolerance, and increases protein turnover.[4] Nutritional deficiencies are in part due to dietary inadequacies, but malabsorption, altered hepatobiliary metabolism, and increased renal excretion can also play a role. Deficiencies of folate and phosphatidylcholine (PC) are often seen with excessive ethanol ingestion.[5] The conversion of methionine to S-adenosylmethionine (SAMe), another crucial methyl donor that can assist in glutathione production, is also inhibited with excessive alcohol intake, particularly when it has led to liver disease.[6] With each of these, the methylation of DNA and other processes of methylation become impaired.
In addition to causing deficiencies of folate, PC, and SAMe, which adversely impact methylation and numerous other chemical reactions, excessive alcohol use can also deplete thiamin (vitamin B1), iron, and pyridoxine (vitamin B6). Studies have shown that up to 80% of binge or chronic heavy drinkers experience thiamin deficiency, more than half experience vitamin B6 deficiency, and two-thirds have folate deficiency.[7],[8],[9] Deficiencies of other water- and fat-soluble vitamins, as well as minerals and trace elements, can also occur, particularly when higher intake contributes to liver and pancreatic disease.[10]
Statistics on specific nutrient deficiencies with the use of addictive substances other than alcohol are harder to come by, as there is often overlapping use of a variety of addictive substances with alcohol. That being said, one study showed that more than 74% of individuals entering treatment for drug addiction had clinical signs of nutrient deficiency, while testing and symptoms showed multiple nutrient deficiencies in more than 60% of individuals.[11] Significantly lower levels of important nutrients with antioxidant potential (vitamins A, C, and E) were also shown in this population, and were not found to be associated with other potential factors such as income and age.[12] Glutathione, the body’s primary intracellular antioxidant that protects the liver from oxidative stress, has been shown to be significantly lower in chronic alcohol users, with or without the presence of cirrhosis.[13],[14]
However, the stress of substance use disorders goes beyond nutritional deficiencies and free radicals, and some of these factors, if neglected, can also have an adverse effect on recovery.[15],[16] Dysregulated function of the hypothalamic-pituitary-adrenal (HPA) axis has been identified in both substance use disorders and other behaviours of addiction.[17],[18],[19],[20] Imbalances in the autonomic nervous system response, both prior and subsequent to alcohol use cessation, have also been documented, and are linked to increased cravings.[21],[22],[23] Heart rate variability (HRV), a marker of autonomic nervous system tone, is reduced in alcohol-dependent individuals, with an even greater reduction in individuals with comorbid symptoms of anxiety and depression.[24] HRV is generally higher with increased parasympathetic tone, suggesting a high sympathetic state in these populations.[25]
The Often-Neglected Stresses in Psychiatric Disorders
Many similar systemic stressors, including nutritional deficiencies, oxidative stress, and HPA axis and autonomic nervous system dysfunction, also exist in psychiatric illnesses. Mood disorders such as anxiety, depression, and bipolar disorder also often come with altered eating patterns, although usually to a lesser extent than substance use disorders. Depression is associated with lower levels of zinc,.[28]
Higher levels of oxidative stress have been shown in psychiatric disorders including anxiety, depression, bipolar disorder, and schizophrenia.[29],[30],[31] Oxidative stress is inherently associated with inflammation, and, perhaps not surprisingly, higher levels of pro-inflammatory cytokines and/or acute-phase reactants such as C-reactive protein (CRP) have been seen in depression,[32],[33] bipolar disorder,[34] and in men and elderly persons with anxiety.[35] Oxidative damage to cellular membranes alters their fluidity (and, with this, functionality) and contributes to mitochondrial dysfunction; these two effects may play a role in the pathogenesis of psychiatric illness.[36],[37],[38] Interestingly, antidepressants have been shown to possibly impact depression by their anti-inflammatory and antioxidative effects.[39],[40]
Hyperactivity of the HPA axis and increased basal cortisol levels are commonly found in patients with depression.[41] Studies have shown a reduced sensitivity of the glucocorticoid receptor at the level of the hypothalamus and pituitary in depressed individuals, leading to impaired negative feedback.[42],[43] In long-standing anxiety disorders, a lower cortisol awakening response has been observed, although findings from studies investigating total cortisol response are inconsistent.[44] Autonomic nervous system dysfunction also is common in psychiatric illness, and HRV has been shown to be reduced across psychiatric disorders compared to healthy controls.[45
Restoring Balance to the Stressed System
There are many important tools that must be implemented when one chooses, or is forced, to cease the abuse of substances. These include psychiatric therapy, acute medical care (if needed), nutritional support, lifestyle changes, and a variety of medications. For many individuals, problems of substance use coexist with mental health struggles, necessitating an individualised approach to treatment. Unfortunately, relapse rates across all types of addiction are high,[46] and the incidence of substance dependency continues to grow.[47] This leads us to question what evidence exists for directed supplement therapies to help promote long-term recovery.
Reducing Free Radicals with Vitamin C and N-Acetylcysteine
Although in clinic we are often faced with an array of options for antioxidant support, there are some very specific antioxidants that should be considered in settings of mental health and addiction. Two primary powerhouses, with evidence backing their use for both mental health and addiction, are vitamin C and N-acetylcysteine (NAC).
Vitamin C is an important cofactor for the synthesis of several hormones and neurotransmitters made in the adrenal gland. Perhaps not surprisingly, the adrenal glands are among the organs with the highest concentration of vitamin C.[48] Ascorbic acid enhances the production of norepinephrine from dopamine,[49] and ascorbic acid deficiency has been shown to be associated with lower levels of dopamine and serotonin metabolites.[50] Studies suggest vitamin C may be useful in settings of stress: multiple animal studies show reduced depressive-like behaviour and oxidative stress when animals are supplemented with vitamin C.[51],[52]
Low plasma ascorbic acid levels have been shown to exist in patients with major depression, as well as in patients entering therapy for drug addiction.10,[53] Vitamin C has been shown to positively impact symptoms of anxiety and depression in multiple randomised, double-blind, placebo-controlled trials (RDBPCTs). It also has been studied in settings of addiction, although RDBPCTs are lacking.
In one RDBPCT, high school students not previously described to have anxiety were evaluated at baseline for heart rate and anxiety with the Beck Anxiety Inventory.[54] Students were then given 500 mg of vitamin C or placebo daily for two weeks. When the same parameters were re-evaluated the day after completing the intervention, it was found that both anxiety and heart rate were significantly reduced in the individuals receiving vitamin C. In another RDBPCT, paediatric patients with major depressive disorder were given 1,000 mg of vitamin C or placebo daily as an adjunctive therapy to the medication fluoxetine.[55] In the children receiving the vitamin C, there was a significant decrease in depressive symptoms as measured by the Children’s Depression Rating Scale and Children’s Depression Inventory. Finally, in a randomised, single-blind study, individuals with type 2 diabetes were given 1,000 mg of vitamin C, 400 IU of vitamin E (alpha-tocopherol form), or placebo daily for six weeks.[56] Patients were evaluated with the Depression Anxiety Stress Scale 21-item questionnaire prior to and at the end of the study. Out of all the groups, the only significant change was a reduction in anxiety for the individuals taking vitamin C.
A buffered ascorbate compound has been studied as a supportive therapy for individuals seeking to detoxify from opiates and stimulants at the Haight-Ashbury Free Medical Clinic.[57] Subjects were given a dosage of buffered vitamin C ranging from 1000 to 3000 mg daily and completed a daily questionnaire to track their detoxification symptoms and substance use. Over half of the clients reported at least 60% relief of their acute withdrawal symptoms, while aftercare clients reported symptom relief of 90%. Of note, it was found that the starting date of the buffered ascorbate compound supplement (during the first 10 days of substance cessation vs. after this time) was not a significant factor in the reported benefits of the therapy, and aftercare clients reported significantly greater benefits than those in active detox. The dosage of buffered ascorbate compound was not found to be a factor, suggesting fairly low dosages could be of benefit. In addition to the benefits of the antioxidants provided by this compound, it has been suggested that the use of alkalinising substances (provided in this compound as well) plays a significant role in promoting effective detoxification.[58]
There also is substantial evidence backing the use of NAC in settings of psychiatric disorders and the cessation of addictive behavior and substance use. NAC itself has antioxidant and anti-inflammatory activity,[59],[60] but possibly more importantly, it provides cysteine, the rate-limiting amino acid for the synthesis of glutathione,[61] the body’s main antioxidant and master detoxifier.[62] NAC has been shown to decrease oxidative stress levels in the body and to reduce homocysteine,[63] which is also often high in individuals with depression and in those who abuse alcohol.[64],[65] Studies have shown that NAC is neuroprotective, crossing the blood–brain barrier and accumulating in the central nervous system;[66] this gives it potential for use in other disorders, including cognitive impairment and traumatic brain injury.[67] NAC has been shown to reduce compulsive behavior via modulation of glutamate, a primary excitatory neurotransmitter that has been demonstrated to be dysregulated in mood disorders and addiction.[68],[69],[70],[71]
Animal and human studies support the use of NAC for the reduction of obsessive-compulsive behaviors. In animal models, NAC has been shown to reduce marble-burying behavior (a typical model used to investigate obsessive-compulsive disorder).[72] Multiple clinical and individual trials in adults and children have been reported, with dosages ranging from 2,400 to 3,000 mg per day, usually for a period of about 12 weeks.[73],[74] Overall, NAC has been shown to reduce the severity of obsessive-compulsive symptoms and be well tolerated.
NAC has also been investigated in multiple clinical trials as an adjunctive treatment for bipolar disorder. At a dosage of 1,000 mg twice daily for 8 to 24 weeks, NAC was shown to significantly reduce the Montgomery-Åsberg Depression Rating Scale[75] and Bipolar Depression Rating Scale scores.[76] A subgroup analysis of patients with bipolar II found that six of seven patients achieved full remission from both depressive and manic symptoms in the NAC group, compared to two of seven in the placebo group.[77] A 2016 systemic review and meta-analysis of the use of NAC for the treatment of depressive symptoms found that in the five studies that qualified for inclusion (including 574 patients), NAC ameliorated depressive symptoms, improved functionality, and was well tolerated.[78]
Given the benefits seen with obsessive-compulsive behaviours and depression, it should not be surprising that an array of studies exists evaluating the potential use of NAC for disorders of addiction. In an open-label outpatient study of cocaine-dependent patients, NAC at dosages of 2,400 or 3,600 mg/day was found to support the termination or reduced use of cocaine.[79] In an additional small, double-blind study in cocaine-dependent patients, NAC at a dose of 2,400 mg/day was found to significantly attenuate the reinforcing effects of cocaine use.[80] In a systemic review of studies related to cocaine addiction, NAC was found in four of the six clinical trials to reduce craving, desire to use cocaine, cocaine-cue viewing time, and cocaine-related spending, with animal models showing a reversal of cocaine use–related disruption of glutamate homeostasis.[81]
In addition to cocaine-use disorder, studies have also looked at the use of NAC for the treatment of smoking cessation, pathological gambling, and alcohol use during cannabis cessation. In a short-term pilot study of smoking cessation, compared to placebo, NAC (at a dosage of 3,600 mg/day) was found to significantly reduce the reward experienced with the first cigarette after a 3.5-day abstinence period.[82] In an open-label study of 27 subjects being treated for gambling addiction, NAC was found to significantly reduce scores related to pathological gambling, with the mean effective dose being 1,476.9 ± 311.3 mg/day.[83] The 16 responders to NAC then entered a double-blind placebo-controlled phase, and of those receiving NAC, 83.3% had improvement of behaviours, compared to 28.6% of those receiving placebo. Finally, in the setting of cannabis cessation (without diagnosed alcohol use disorders, and having no desire to alter alcohol habits), treatment with 1,200 mg of NAC twice daily was found to increase the odds of simultaneous alcohol abstinence, reduce weekly alcohol use, and decrease drinking days.[84]
Rebalancing the HPA Axis and Parasympathetic Nervous System with Ashwagandha and Phosphatidylserine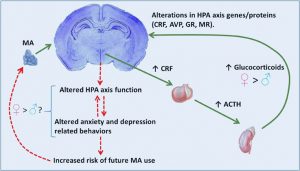
In addition to addressing the stress on the system in settings of mental health and substance abuse from the perspective of antioxidant therapies, it also is imperative to address the stress-mediating activity of the HPA axis and parasympathetic nervous system dysregulation. Alterations in the HPA axis response are common with addiction and have been shown to be associated with shorter time to relapse.[85],[86],[87] Dysregulation of the HPA axis may be a major contributor to insomnia, which is a robust predictor of increased relapse rates for alcohol use[88] and possibly other substances[89],[90] On the parasympathetic nervous system side, alcohol promotes GABA release from the central amygdala,[91] a factor which plays a role in reinforcing patterns of alcohol use, particularly for anxiety-driven drinking patterns.[92],[93] GABA agonists have thus been proposed as a clinical target for the treatment of both alcohol and substance use disorders.[94]
Indian ginseng (Withania somnifera), commonly known as ashwagandha, has been studied in a wide array of clinical and animal studies. Ashwagandha is considered a nerve tonic and adaptogen; that is, an herb that has been shown to help modulate the body’s response to physical, mental, and emotional stressors. A variety of mechanisms by which ashwagandha can help mediate substance use patterns, improve mental health, and support healthy sleep have been demonstrated in numerous clinical settings. Ashwagandha has been shown to blunt the stress-induced activation of the HPA axis[95] and to interact with GABA receptors.[96],[97] The Latin naming of ashwagandha, somnifera, also reflects the ability of this herb to induce sleep,[98] which may be mediated by both GABAergic signaling and regulation of HPA axis activity. GABAergic signaling is also involved in mediating pain,[99] which ashwagandha has been shown to reduce in humans as well.[100]
Ashwagandha has an array of clinical evidence in support of its use for mental health. A RDBPCT in patients with obsessive-compulsive disorder showed that 120 mg of ashwagandha daily (in addition to ongoing treatment with selective serotonin reuptake inhibitors) significantly improved symptoms as assessed by the Yale-Brown Obsessive Compulsive Scale when reassessed at six weeks.[101] Multiple clinical trials have shown that ashwagandha reduces anxiety,[102],[103] with a systemic review showing significant benefit of ashwagandha over placebo.[104] Although ashwagandha has not been studied in clinical settings of substance use and addiction, animal models have shown that it helps to reduce alcohol withdrawal–associated symptoms,
Phosphatidylserine (PS) is another substance that has been shown to regulate the HPA axis response to stress.[107] PS is a phospholipid found in the body’s cellular membranes, including the neurons and the myelin that protects them both centrally and peripherally.[108] In the brain, PS plays an important role in neurotransmitter release and cellular signaling.[109] PS has also been shown to stimulate GABA uptake in neuronal cells.[110],[111] Ethanol inhibits neuronal accumulation of PS, contributing to cellular apoptosis and possibly deficits in brain function.[112] Essential fatty acid deficiencies further contribute to diminished levels of obligatory PS in the neuronal membranes.[113]
Multiple clinical studies have demonstrated the impact PS has on the stress-related response as well as mood. PS has been shown to modulate the HPA axis response to both physical and social stressors; after a short period of supplementation, PS reduces the typical rise in both adrenocorticotropic hormone (ACTH) and cortisol from these typical stressors.99,[114] In a RDBPCT with a population of males under chronic stress, a combination of PS with phosphatidic acid (PA), another phospholipid which comprises much of the cellular membrane, was shown to significantly normalize the ACTH and cortisol response in those under chronically higher levels of stress than the group as a whole.[115] Improvements in mood have been seen in both healthy populations and those with mood-related issues. After one month of supplementation of 300 mg of PS daily, healthy young adults reported feeling less stressed and being in a better mood.[116] Mood-related benefits of PS supplementation also have been seen in elderly women with depressive disorders (along with improvements in memory)[117] and women with premenstrual syndrome.[118]
Although the challenges when supporting patients with mental health issues and addiction are vast, vitamin C, NAC, ashwagandha, and PS each have many potential mechanisms by which they can help. Clinical evidence justifies their use on both a short- and a long-term basis in supporting these populations.
References
[1] Santolaria F, et al. Nutritional assessment in alcoholic patients. Its relationship with alcoholic intake, feeding habits, organic complications and social problems. Drug Alcohol Depend. 2000 Jun 1;59(3):295-304.
[2] Salaspuro M. Nutrient intake and nutritional status in alcoholics. Alcohol Alcohol. 1993 Jan;28(1):85-8.
[3] Moreno Otero R, Cortés JR. [Nutrition and chronic alcohol abuse]. Nutr Hosp. 2008 May;23 Suppl 2:3-7.
[4] Bunout D. Nutritional and metabolic effects of alcoholism: their relationship with alcoholic liver disease. Nutrition. 1999 Jul-Aug;15(7-8):583-9.
[5] Hamid A, et al. New perspectives on folate transport in relation to alcoholism-induced folate malabsorption–association with epigenome stability and cancer development. FEBS J. 2009 Apr;276(8):2175-91.
[6] Lieber CS. ALCOHOL: its metabolism and interaction with nutrients. Annu Rev Nutr. 2000;20:395-430.
[7] Cabré E, Gassull MA. Nutritional aspects of liver disease and transplantation. Curr Opin Clin Nutr Metab Care. 2001 Nov;4(6):581-9.
[8] Markowitz JS, et al. Oral nutritional supplementation for the alcoholic patient: a brief overview. Ann Clin Psychiatry. 2000;12(3):153.
[9] Fonda ML, et al. Concentration of vitamin B6 and activities of enzymes of B6 metabolism in the blood of alcoholic and nonalcoholic men. Alcohol Clin Exp Res. 1989;13(6):804.
[10] Leevy CM, Moroianu SA. Nutritional aspects of alcoholic liver disease. Clin Liver Dis. 2005;9(1):67.
[11] Nazrul Islam SK, et al. Nutritional status of drug addicts undergoing detoxification: prevalence of malnutrition and influence of illicit drugs and lifestyle. Br J Nutr. 2002 Nov;88(5):507-13.
[12] Nazrul Islam SK, et al. Serum vitamin E, C and A status of the drug addicts undergoing detoxification: influence of drug habit, sexual practice and lifestyle factors. Eur J Clin Nutr. 2001 Nov;55(11):1022-7.
[13] Loguercio C, et al. Ethanol consumption, amino acid and glutathione blood levels in patients with and without chronic liver disease. Alcohol Clin Exp Res. 1999 Nov;23(11):1780-4.
[14] Loguercio C, et al. Determination of plasma alpha-glutathione-S-transferases in chronic alcohol abusers: relationship with alcohol intake and liver involvement. Alcohol Alcohol. 1998 Jul-Aug;33(4):366-72.
[15] Adinoff B, et al. Suppression of the HPA axis stress-response: implications for relapse. Alcohol Clin Exp Res. 2005 Jul;29(7):1351-5.
[16] Sinha R, et al. Stress-induced cocaine craving and hypothalamic-pituitary-adrenal responses are predictive of cocaine relapse outcomes. Arch Gen Psychiatry. 2006 Mar;63(3):324-31.
[17] Chatzittofis A, et al. HPA axis dysregulation in men with hypersexual disorder. Psychoneuroendocrinology. 2016 Jan;63:247-53.
[18] Goeders NE. Stress and cocaine addiction. J Pharmacol Exp Ther. 2002 Jun;301(3):785-9.
[19] Geisel O, et al. Hypothalamic-pituitary-adrenal axis activity in patients with pathological gambling and internet use disorder. Psychiatry Res. 2015 Mar 30;226(1):97-102.
[20] Costa A, et al. An assessment of hypothalamo-pituitary-adrenal axis functioning in non-depressed, early abstinent alcoholics. Psychoneuroendocrinology. 1996 Apr;21(3):263-75.
[21] Yokoyama A, et al. Impaired autonomic nervous system in alcoholics assessed by heart rate variation. Alcohol Clin Exp Res. 1991 Oct;15(5):761-5.
[22] de Zambotti M, et al. Poor autonomic nervous system functioning during sleep in recently detoxified alcohol-dependent men and women. Alcohol Clin Exp Res. 2014 May;38(5):1373-80.
[23] Penzlin AI, et al. Heart rate variability biofeedback in patients with alcohol dependence: a randomized controlled study. Neuropsychiatr Dis Treat. 2015 Oct 9;11:2619-27.
[24] Chen TY, et al. Different patterns of heart rate variability during acute withdrawal in alcohol dependent patients with and without comorbid anxiety and/or depression. J of Psychophys. 2015 Jul 31;29:87-98.
[25] Goldberger JJ, et al. Relationship of heart rate variability to parasympathetic effect. Circulation. 2001 Apr 17;103(15):1977-83.
[26] Petrilli MA, et al. The Emerging Role for Zinc in Depression and Psychosis. Front Pharmacol. 2017 Jun 30;8:414.
[27] Coppen A, Bolander-Gouaille C. Treatment of depression: time to consider folic acid and vitamin B12. J Psychopharmacol. 2005 Jan;19(1):59-65.
[28] Carney MW, et al. Thiamine, riboflavin and pyridoxine deficiency in psychiatric in-patients. Br J Psychiatry. 1982 Sep;141:271-2.
[29] Moylan S, et al. Oxidative & nitrosative stress in depression: why so much stress? Neurosci Biobehav Rev. 2014;45:46-62.
[30] Ng F, et al. Oxidative stress in psychiatric disorders: evidence base and therapeutic implications. Int J Neuropsychopharmacol. 2008 Sep;11(6):851-76.
[31] Smaga I, et al. Oxidative stress as an etiological factor and a potential treatment target of psychiatric disorders. Part 2. Depression, anxiety, schizophrenia and autism. Pharmacol Rep. 2015 Jun;67(3):569-80.
[32] Pasco JA, et al. Association of high-sensitivity C-reactive protein with de novo major depression. Br J Psychiatry. 2010;197(5):372-7.
[33] Dowlati Y, et al. A meta-analysis of cytokines in major depression. Biol Psychiatry. 2010;67(5):446-57.
[34] Kim YK, et al. Imbalance between pro-inflammatory and anti-inflammatory cytokines in bipolar disorder. J Affect Disord. 2007 Dec;104(1-3):91-5.
[35] Vogelzangs N, et al. Anxiety disorders and inflammation in a large adult cohort. Transl Psychiatry. 2013 Apr 23;3:e249.
[36] Tsuboi H, et al. Depressive symptoms are independently correlated with lipid peroxidation in a female population: comparison with vitamins and carotenoids. J Psychosom Res. 2004 Jan;56(1):53-8.
[37] Jou SH, et al. Mitochondrial dysfunction and psychiatric disorders. Chang Gung Med J. 2009;32(4):370-9.
[38] Allen J, et al. Mitochondria and Mood: Mitochondrial Dysfunction as a Key Player in the Manifestation of Depression. Front Neurosci. 2018 Jun 6;12:386.
[39] Dinan TG. Inflammatory markers in depression. Curr Opin Psychiatry. 2009;22(1):32-6.
[40] Caiaffo V, et al. Anti-inflammatory, antiapoptotic, and antioxidant activity of fluoxetine. Pharmacol Res Perspect. 2016;4(3):e00231.
[41] Pariante CM, Lightman SL. The HPA axis in major depression: classical theories and new developments. Trends Neurosci. 2008;31(9):464-8.
[42] Coppen A, et al. Dexamethasone suppression test in depression and other psychiatric illness. Br J Psychiatry. 1983;142:498-504.
[43] Lopez-Duran NL, et al. Hypothalamic-pituitary-adrenal axis dysregulation in depressed children and adolescents: a meta-analysis. Psychoneuroendocrinology. 2009;34(9):1272-83.
[44] Hek K, et al. Anxiety disorders and salivary cortisol levels in older adults: a population-based study. Psychoneuroendocrinology. 2013;38(2):300-5.
[45] Alvares GA, et al. Autonomic nervous system dysfunction in psychiatric disorders and the impact of psychotropic medications: a systematic review and meta-analysis. J Psychiatry Neurosci. 2016 Mar;41(2):89-104.
[46] Milkman H, et al. Addiction relapse. Adv Alcohol Subst Abuse. 1983-1984 Fall-Winter;3(1-2):119-34.
[47] Degenhardt L, et al. Global burden of disease attributable to illicit drug use and dependence: findings from the Global Burden of Disease Study 2010. Lancet. 2013 Nov 9;382(9904):1564-74.
[48] Patak P, et al. Vitamin C is an important cofactor for both adrenal cortex and adrenal medulla. Endocr Res. 2004 Nov;30(4):871-5.
[49] May JM, et al. Ascorbic acid efficiently enhances neuronal synthesis of norepinephrine from dopamine. Brain Res Bull. 2013 Jan;90:35-42.
[50] Ward MS, et al. Behavioral and monoamine changes following severe vitamin C deficiency. J Neurochem. 2013 Feb;124(3):363-75.
[51] Moretti M, et al. Ascorbic acid treatment, similarly to fluoxetine, reverses depressive-like behavior and brain oxidative damage induced by chronic unpredictable stress. J Psychiatr Res. 2012 Mar;46(3):331-40.
[52] Moretti M, et al. Protective effects of ascorbic acid on behavior and oxidative status of restraint-stressed mice. J Mol Neurosci. 2013 Jan;49(1):68-79.
[53] Khanzode SD, et al. Oxidative damage and major depression: the potential antioxidant action of selective serotonin re-uptake inhibitors. Redox Rep. 2003;8(6):365-70.
[54] de Oliveira IJ, et al. Effects of Oral Vitamin C Supplementation on Anxiety in Students: A Double-Blind, Randomized, Placebo-Controlled Trial. Pak J Biol Sci. 2015 Jan;18(1):11-8.
[55] Amr M, et al. Efficacy of vitamin C as an adjunct to fluoxetine therapy in pediatric major depressive disorder: a randomized, double-blind, placebo-controlled pilot study. Nutr J. 2013 Mar;12(1):1.
[56] Mazloom Z, et al. Efficacy of supplementary vitamins C and E on anxiety, depression and stress in type 2 diabetic patients: a randomized, single-blind, placebo-controlled trial. Pak J Biol Sci. 2013 Nov 15;16(22):1597-600.
[57] Newmeyer J, et al. Efficacy of Buffered Ascorbate Compound (BAC) in the Detoxification and Aftercare of Clients Involved in Opiate and Stimulant Abuse. From the Haight-Ashbury Free Medical Clinic. 1983 Jul:1-5.
[58] Minich DM, Bland JS. Acid-alkaline balance: role in chronic disease and detoxification. Altern Ther Health Med. 2007 Jul-Aug;13(4):62-5.
[59] Samuni Y, et al. The chemistry and biological activities of N-acetylcysteine. Biochim Biophys Acta. 2013 Aug;1830(8):4117-29.
[60] Pei Y, et al. Biological Activities and Potential Oral Applications of N-Acetylcysteine: Progress and Prospects. Oxid Med Cell Longev. 2018 Apr 22;2018:2835787.
[61] Rushworth GF, Megson IL. Existing and potential therapeutic uses for N-acetylcysteine: the need for conversion to intracellular glutathione for antioxidant benefits. Pharmacol Ther. 2014 Feb;141(2):150-9.
[62] Devasagayam TP, et al. Free radicals and antioxidants in human health: current status and future prospects. J Assoc Physicians India. 2004 Oct;52:794-804.
[63] Kasperczyk S, et al. Effect of N-acetylcysteine administration on homocysteine level, oxidative damage to proteins, and levels of iron (Fe) and Fe-related proteins in lead-exposed workers. Toxicol Ind Health. 2016 Sep;32(9):1607-18.
[64] Nabi H, et al. Association of serum homocysteine with major depressive disorder: results from a large population-based study. Psychoneuroendocrinology. 2013 Oct;38(10):2309-18.
[65] Bleich S, et al. Evidence of increased homocysteine levels in alcoholism: the Franconian alcoholism research studies (FARS). Alcohol Clin Exp Res. 2005 Mar;29(3):334-6.
[66] Farr SA, et al. The antioxidants alpha-lipoic acid and N-acetylcysteine reverse memory impairment and brain oxidative stress in aged SAMP8 mice. J Neurochem. 2003 Mar;84(5):1173-83.
[67] Bhatti J, et al. Systematic Review of Human and Animal Studies Examining the Efficacy and Safety of N-Acetylcysteine (NAC) and N-Acetylcysteine Amide (NACA) in Traumatic Brain Injury: Impact on Neurofunctional Outcome and Biomarkers of Oxidative Stress and Inflammation. Front Neurol. 2018 Jan 15;8:744.
[68] Chakrabarty K, et al. Glutamatergic dysfunction in OCD. Neuropsychopharmacology. 2005 Sep;30(9):1735-40.
[69] Müller N, Schwarz MJ. The immune-mediated alteration of serotonin and glutamate: towards an integrated view of depression. Mol Psychiatry. 2007 Nov;12(11):988-1000.
[70] Terbeck S, et al. The role of metabotropic glutamate receptor 5 in the pathogenesis of mood disorders and addiction: combining preclinical evidence with human Positron Emission Tomography (PET) studies. Front Neurosci. 2015 Mar 18;9:86.
[71] Kalivas PW, et al. The glutamate homeostasis hypothesis of addiction. Nat Rev Neurosci. 2009 Aug;10(8):561-72.
[72] Egashira N, et al. N-Acetyl-L-Cysteine Inhibits Marble-Burying Behavior in Mice. J Pharmacol Sci. 2012;119(1):97-101.
[73] Oliver G, et al. N-acetyl cysteine in the treatment of obsessive compulsive and related disorders: a systematic review. Clin Psychopharmacol Neurosci. 2015 Apr 30;13(1):12-24.
[74] Couto JP, Moreira R. Oral N-acetylcysteine in the treatment of obsessive-compulsive disorder: A systematic review of the clinical evidence. Prog Neuropsychopharmacol Biol Psychiatry. 2018 Aug 30;86:245-54.
[75] Berk M, et al. N-acetyl cysteine for depressive symptoms in bipolar disorder–a double-blind randomized placebo-controlled trial. Biol Psychiatry. 2008 Sep 15;64(6):468-75.
[76] Berk M, et al. The efficacy of N-acetylcysteine as an adjunctive treatment in bipolar depression: an open label trial. J Affect Disord. 2011 Dec;135(1-3):389-94.
[77] Magalhães PV, et al. N-acetyl cysteine add-on treatment for bipolar II disorder: a subgroup analysis of a randomized placebo-controlled trial. J Affect Disord. 2011 Mar;129(1-3):317-20.
[78] Fernandes BS, et al. N-Acetylcysteine in depressive symptoms and functionality: a systematic review and meta-analysis. J Clin Psychiatry. 2016 Apr;77(4):e457-66.
[79] Mardikian PN, et al. An open-label trial of N-acetylcysteine for the treatment of cocaine dependence: a pilot study. Prog Neuropsychopharmacol Biol Psychiatry. 2007 Mar 30;31(2):389-94.
[80] Levi Bolin B, et al. N-Acetylcysteine reduces cocaine-cue attentional bias and differentially alters cocaine self-administration based on dosing order. Drug Alcohol Depend. 2017 Sep 1;178:452-60.
[81] Nocito Echevarria MA, et al. N-acetylcysteine for treating cocaine addiction – A systematic review. Psychiatry Res. 2017 May;251:197-203.
[82] Schmaal L, et al. Efficacy of N-acetylcysteine in the treatment of nicotine dependence: a double-blind placebo-controlled pilot study. Eur Addict Res. 2011;17(4):211-6.
[83] Grant JE, et al. N-acetyl cysteine, a glutamate-modulating agent, in the treatment of pathological gambling: a pilot study. Biol Psychiatry. 2007 Sep 15;62(6):652-7.
[84] Squeglia LM, et al. The effect of N-acetylcysteine on alcohol use during a cannabis cessation trial. Drug Alcohol Depend. 2018 Apr 1;185:17-22.
[85] Sinha R, et al. Effects of adrenal sensitivity, stress- and cue-induced craving, and anxiety on subsequent alcohol relapse and treatment outcomes. Arch Gen Psychiatry. 2011 Sep;68(9):942-52.
[86] al’Absi M. Hypothalamic-pituitary-adrenocortical responses to psychological stress and risk for smoking relapse. Int J Psychophysiol. 2006 Mar;59(3):218-27.
[87] Junghanns K, et al. Impaired serum cortisol stress response is a predictor of early relapse. Alcohol Alcohol. 2003 Mar-Apr;38(2):189-93.
[88] Brower KJ, et al. Insomnia, self-medication, and relapse to alcoholism. Am J Psychiatry. 2001 Mar;158(3):399-404.
[89] Brower KJ, Perron BE. Sleep disturbance as a universal risk factor for relapse in addictions to psychoactive substances. Med Hypotheses. 2010 May;74(5):928-33.
[90] Roehrs TA, Roth T. Sleep Disturbance in Substance Use Disorders. Psychiatr Clin North Am. 2015 Dec;38(4):793-803.
[91] Roberto M, et al. Corticotropin releasing factor-induced amygdala gamma-aminobutyric Acid release plays a key role in alcohol dependence. Biol Psychiatry. 2010 May 1;67(9):831-9.
[92] Koob GF. A role for GABA mechanisms in the motivational effects of alcohol. Biochem Pharmacol. 2004 Oct 15;68(8):1515-25.
[93] Augier E, et al. The GABAB Positive Allosteric Modulator ADX71441 Attenuates Alcohol Self-Administration and Relapse to Alcohol Seeking in Rats. Neuropsychopharmacology. 2017 Aug;42(9):1789-99.
[94] Agabio R, Colombo G. [GABAB receptor as therapeutic target for drug addiction: from baclofen to positive allosteric modulators]. Psychiatr Pol. 2015 Mar-Apr;49(2):215-23.
[95] Chandrasekhar K, et al. A prospective, randomized double-blind, placebo-controlled study of safety and efficacy of a high-concentration full-spectrum extract of ashwagandha root in reducing stress and anxiety in adults. Indian J Psychol Med. 2012 Jul;34(3):255-62.
[96] Candelario M, et al. Direct evidence for GABAergic activity of Withania somnifera on mammalian ionotropic GABAA and GABAρ receptors. J Ethnopharmacol. 2015 Aug 2;171:264-72.
[97] Yin H, et al. GABA-mimetic actions of Withania somnifera on substantia gelatinosa neurons of the trigeminal subnucleus caudalis in mice. Am J Chin Med. 2013;41(5):1043-51.
[98] Kaushik MK, et al. Triethylene glycol, an active component of Ashwagandha (Withania somnifera) leaves, is responsible for sleep induction. PLoS One. 2017 Feb 16;12(2):e0172508.
[99] Enna SJ, McCarson KE. The role of GABA in the mediation and perception of pain. Adv Pharmacol. 2006;54:1-27.
[100] Nalini P, et al. Evaluation of the analgesic activity of standardized aqueous extract of Withania somnifera in healthy human volunteers using Hot Air Pain Model. Res J Life Sci. 2013;1(2):1-6.
[101] Jahanbakhsh SP, et al. Evaluation of the efficacy of Withania somnifera (Ashwagandha) root extract in patients with obsessive-compulsive disorder: A randomized double-blind placebo-controlled trial. Complement Ther Med. 2016 Aug;27:25-9.
[102] SudKhyati S, Anup BT. A randomized double-blind placebo controlled study of ashwagandha on generalized anxiety disorder. Int Ayurvedic Med J. 2013;1:1-7.
[103] Auddy B, et al. A standardized Withania somnifera extract significantly reduces stress-related parameters in chronically stressed humans; A double-blind, randomized, placebo-controlled study. JANA. 2008; 11:50-6.
[104] Pratte MA, et al. An alternative treatment for anxiety: a systematic review of human trial results reported for the Ayurvedic herb ashwagandha (Withania somnifera). J Altern Complement Med. 2014 Dec;20(12):901-8.
[105] Ruby B, et al. Evaluation of ashwagandha in alcohol withdrawal syndrome. Asian Pac J Trop Dis. 2012 Jan 1;2:S856-60.
[106] Bansal P, Banerjee S. Effect of Withinia Somnifera and Shilajit on Alcohol Addiction in Mice. Pharmacogn Mag. 2016 May;12(Suppl 2):S121-8.
[107] Monteleone P, et al. Blunting by chronic phosphatidylserine administration of the stress-induced activation of the hypothalamo-pituitary-adrenal axis in healthy men. Eur J Clin Pharmacol. 1992;42(4):385-8.
[108] Glade MJ, Smith K. Phosphatidylserine and the human brain. Nutrition. 2015 Jun;31(6):781-6.
[109] Kim HY, et al. Phosphatidylserine in the brain: metabolism and function. Prog Lipid Res. 2014 Oct;56:1-18.
[110] Chweh AY, Leslie SW. Phosphatidylserine enhancement of [3H]gamma-aminobutyric acid uptake by rat whole brain synaptosomes. J Neurochem. 1982 Mar;38(3):691-5.
[111] Loeb C, et al. Phosphatidylserine increases in vivo the synaptosomal uptake of exogenous GABA in rats. Exp Neurol. 1988 Feb;99(2):440-6.
[112] Akbar M, et al. Ethanol promotes neuronal apoptosis by inhibiting phosphatidylserine accumulation. J Neurosci Res. 2006 Feb 15;83(3):432-40.
[113] Hamilton L, et al. n-3 fatty acid deficiency decreases phosphatidylserine accumulation selectively in neuronal tissues. Lipids. 2000 Aug;35(8):863-9.
[114] Hellhammer J, et al. Effects of soy lecithin phosphatidic acid and phosphatidylserine complex (PAS) on the endocrine and psychological responses to mental stress. Stress. 2004 Jun;7(2):119-26.
[115] Hellhammer J, et al. A soy-based phosphatidylserine/ phosphatidic acid complex (PAS) normalizes the stress reactivity of hypothalamus-pituitary-adrenal-axis in chronically stressed male subjects: a randomized, placebo-controlled study. Lipids Health Dis. 2014 Jul 31;13:121.
[116] Benton D, et al. The influence of phosphatidylserine supplementation on mood and heart rate when faced with an acute stressor. Nutr Neurosci. 2001;4(3):169-78.
[117] Maggioni M, et al. Effects of phosphatidylserine therapy in geriatric patients with depressive disorders. Acta Psychiatr Scand. 1990 Mar;81(3):265-70.
[118] Schmidt K, et al. A lecithin phosphatidylserine and phosphatidic acid complex (PAS) reduces symptoms of the premenstrual syndrome (PMS): Results of a randomized, placebo-controlled, double-blind clinical trial. Clin Nutr ESPEN. 2018 Apr;24:22-30.






1 Comment. Leave new
Thank you for all the Amazing information in this article.