 Todd A. Born is a naturopathic physician, certified nutrition specialist (CNS), and co-owner and medical director of Born Integrative Medicine Specialists, PLLC. His roles at Allergy Research Group include Product Manager, Head of New Product Development, Scientific Advisor, and Editor-in-Chief of their science-based FOCUS Newsletter. Dr. Born is also Lead Advisor and President of the International Society for Naturopathic Medicine, as well as a medical wellness advisor for the International Medical Wellness Association.
Todd A. Born is a naturopathic physician, certified nutrition specialist (CNS), and co-owner and medical director of Born Integrative Medicine Specialists, PLLC. His roles at Allergy Research Group include Product Manager, Head of New Product Development, Scientific Advisor, and Editor-in-Chief of their science-based FOCUS Newsletter. Dr. Born is also Lead Advisor and President of the International Society for Naturopathic Medicine, as well as a medical wellness advisor for the International Medical Wellness Association.
A review of the evidence behind botanicals and nutraceuticals for the treatment of mood disorders
Mood disorders are mental health issues that primarily affect a person’s emotional state; in these disorders, a person experiences long periods of extreme happiness, extreme sadness, or both.[1] In the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5), the list of mood disorders and their diagnostic criteria are exceedingly lengthy, but the most common include major depression; dysthymia; bipolar disorder; seasonal affective disorder (SAD); and depression related to an illness, substance abuse, or medication.[2] For simplicity, in this article, I will refer to these issues collectively as mood disorders, unless otherwise indicated.
The 2001 to 2003 National Comorbidity Survey Replication shows an estimated 9.7% of U.S. adults (anyone over 18) had any mood disorder in the past year, with a higher prevalence in females (11.6%) than males (7.7%), and an estimated 21.4% of U.S. adults will experience a mood disorder at some point in their lives.[3] (Please note that these figures are a combination of mood disorders; frequently updated individual statistics may be found at “Statistics” on the National Institute of Mental Health website at www.nimh.nih.gov/health/statistics/index.shtml).
Depression alone affects more than 300 million people worldwide.[4] Bipolar disorder affects more than 60 million, and SAD can affect up to 10% of the population, depending on geographical location.[5] The direct and indirect costs of these health issues are immense. For example, in 2008, the World Health Organisation (WHO) ranked major depression as the third leading cause of burden of disease worldwide and projected that the disease will rank first by 2030.[6] In 2015, it was estimated that the total global cost of treatment for Bipolar I Disorder (BDI) was $202.1 billion USD, an average of $81,559 USD per affected individual.
Sadly, the aforementioned numbers of afflicted individuals, as well as cost burden, are likely underestimated because many of the individuals that struggle with mood disorders go undiagnosed. At times, they may be reluctant to seek help due to the social stigma associated with these conditions. In practice, mood disorder detection, diagnosis, and management also pose many challenges for clinicians because of various presentations, unpredictable course and prognosis, as well as variable response to treatment.[7],[8]
There is an urgent need for safer and more efficacious alternatives to medications. Medications certainly have their role in mood disorders, but they must be used judiciously. For example, it has been well established that, in mild to moderate depression, medications are no more effective than placebo.[9] In manic and depressive states of bipolar disorder, the data is mixed on how efficacious medications really are.[10]
The extensive laundry list of side effects of mood disorder medications is so vast, it makes one’s head spin. In clinical practice, it seems the side effects tend to be so intolerable for patients, they would often rather suffer from their illness than deal with the negative consequences of the medications. Antidepressants, anxiolytics, stimulants, antipsychotics, and mood stabilizers are the medications typically used with mood disorders, but a full discussion of their efficacy and potential side effects is beyond the scope of this article.
To better understand mood disorders and provide more effective treatment interventions, it is beneficial to look at underlying aetiologies, risk factors, and genetics (encompassing family history). Each particular mood disorder has its own unique set of aetiologies, but commonalities among all of them include brain structural changes (not yet well understood) compared to those without mood disorders, neurotransmitter alterations, and hormonal imbalances. Risk factors include low self-esteem, being highly self-critical, experiencing traumatic and stressful events (e.g., physical or sexual abuse or loss of a loved one), comorbid mental disorder (e.g., schizophrenia or anxiety disorders), alcohol and recreational drug abuse, and chronic illness and side effects of certain medications (e.g., sleeping pills and antihypertensive drugs).[11]
An enormous risk factor is one’s genetics[12] and even epigenetics.[13] Research into the role single nucleotide polymorphisms (SNPs) play in mood disorders has exploded in the last 15 years or so and offers promise to help improve people’s lives through nutritional and botanical interventions.[14],[15] Indeed, even pharmacogenetic testing is finally becoming more mainstream in clinical practice, offering safer, more specific, and personalised pharmaceutical options.[16],[17],[18]
A comprehensive, integrative approach to mood disorders works very well in clinical practice.[19] This may include all, or most of the following: lifestyle and dietary modifications, constitutional homeopathy, botanical medicines, nutraceutical support, psychotherapy, and occasionally pharmaceutical interventions. Again, given space limitations, this article will focus on evidence-based botanical and nutraceutical interventions.
Studies are consistent that up to half of all individuals diagnosed with a mood disorder use one or more complementary and alternative medicine (CAM) therapies.[20],[21] It has been shown that healthy diet,[22] exercise,[23] sleep; a strong supportive social network; and a low-stress environment reduce relapses in mood disorders.[24] Even targeting the proverbial “gut microbiome” can have a tremendous positive impact![25],[26]
Given all of the aforementioned information, it’s clear there is a dire need for safe and effective alternatives. But are there really any nutraceutical and/or botanical interventions that work? The answer is: yes, definitely.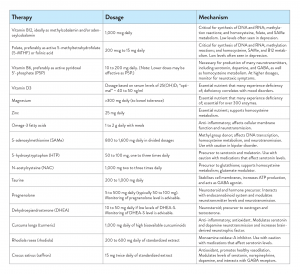
Vitamins and Minerals
Vitamin B12 (cobalamin) is involved in DNA synthesis, red blood cell formation, homocysteine metabolism, and synthesis of S-adenosylmethionine (SAMe). It is also heavily involved in the proper function of the nervous and immune systems.[27] Observational studies have shown that as many as 30% of patients hospitalised with depression are deficient in this vitamin.[28] Depression can be induced by B12 deficiency, even with normal hematological and blood parameters,[29] so a therapeutic intervention of 1,000 mcg (1 mg) daily, orally, has been suggested.[30] The forms of B12 in these studies have varied.
It has been shown that individuals with psychiatric conditions either have impaired transport across the blood–brain barrier and/or an accelerated catabolism, hence the need for increased requirements.[31] When these individuals were treated with a therapeutic trial of B12, clinical improvement was noted.[32]
Folate is a generic term referring to both natural folates in food and folic acid (the synthetic form used in many supplements and fortified food). Folate is critical for the synthesis of DNA and RNA, several amino acids, methylation reactions, homocysteine, and B12 metabolism, and it assists in the proper functioning of the central nervous and immune systems.[33]
Like B12, low red blood cell folate levels have been detected in 15 to 38% of adults diagnosed with depressive disorders.[34] Efficacious doses have ranged from 200 mcg to 15 mg of folic acid, along with medication(s), depending on the mood disorder.[35],[36] Do note, most trials have been conducted on folic acid, not its biologically active forms of 5-methyltetrahydrofolate (5-MTHF, the major circulating form of folate in the body) and 5,10-methylenetetrahydrofolate. In individuals with methylenetetrahydrofolate reductase (MTHFR) polymorphisms and/or who are on medications that inhibit dihydrofolate reductase (by reducing interactions), along with individuals having compromised gastrointestinal function, folinic acid and 5-MTHF may be the preferred forms.[37],[38]
Vitamin B6 (pyridoxine, pyridoxal, and pyridoxamine) and its coenzyme form, pyridoxal 5′-phosphate (P5P), are essential to over 100 enzymes, affecting lipid, amino acid, and carbohydrate metabolism, along with action of steroid hormones.[39] It cannot be synthesized in the body and must be obtained from the diet. In the brain, P5P is necessary to metabolise serotonin from tryptophan and dopamine from L-3,4-dihydroxyphenylalanine (L-Dopa). Other neurotransmitters and amino acids that are P5P dependent include glycine, D-serine, glutamate, histamine, and gamma-aminobutyric acid (GABA).[40] P5P also plays a role in the metabolism of homocysteine.[41] Typical dose ranges are from 10 to 200 mg/day, but anyone taking more than 200 mg/day should be monitored for neurotoxic symptoms.[42]
Vitamin D3 (cholecalciferol) is a fat-soluble vitamin that functions as a hormone precursor. It is biologically inactive and must first be hydroxylated in the liver to 25-hydroxyvitamin D (25[OH]D), with further hydroxylation in the kidneys to its active form, 1,25-dihydroxyvitamin D, the form that acts as a steroid. In this form, it suppresses prostaglandin action; inhibits p38 stress kinase signaling, tumor angiogenesis, invasion and metastasis; and inhibits nuclear factor-kappa B (NF-κB) signaling.[43],[44]
Many studies have seen a correlation between low serum concentrations of 25(OH)D and mood disorders.[45] While the exact mechanism(s) of action haven’t been fully elucidated, vitamin D supplementation has been shown to improve mood in both depression and SAD.[46],[47] It should be noted that “optimal” serum levels of 25(OH)D are around 40 ng/mL.[48],[49]
Magnesium insufficiency and frank deficiency are rampant in the U.S. and most industrialised nations. More than half of the U.S. population (ages ≥4 years) is considered to be underconsuming this vital mineral.[50] Chronic diseases, medications, decreases in food crop magnesium contents, and the availability of refined and processed foods[51] have all contributed to this epidemic.
Magnesium is the second most abundant cation in soft tissues (behind potassium), and it is a cofactor for more than 300 enzymes. It plays a role in adenosine triphosphate (ATP) production, neuronal activity, cardiac function, and electrical properties of cell membranes; has antispasmodic effects; and assists in glutathione synthesis.[52],[53] In addition to all of these accolades, magnesium has anxiolytic properties, increases stress tolerance, and is a great antidepressant.[54],[55],[56] The recommended daily allowance (RDA) varies by age and gender, ranging from 360 to 420 mg of elemental magnesium daily.
Zinc, well known for its immune properties, also plays an important role in mood, as it is essential for over 300 enzyme-dependent reactions.[57] The RDA for individuals 19 years and older is 11 mg daily for males and 8 mg daily for females. Therapeutically, trials have shown efficacy at much higher doses, although this depends on the condition being addressed. In many of the depression trials, a dose of 25 mg daily of elemental zinc was utilized.[58],[59] Excessive zinc intake can have toxic effects, as well as deplete copper, so the U.S. Food and Nutrition Board has set the tolerable upper limit for those 19 years or older at 40 mg/day.[60
Omega-3 Fatty Acids
Eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) are well known for anti-inflammatory affects[61],[62] via suppression of NF-κB, cyclooxygenase-2 (COX-2), tumor necrosis factor alpha (TNFα), and interleukin beta (IL-1β). They also have a solid reputation for assisting those afflicted with mood disorders. Doses of 500 mg to 10 g of EPA and DHA, with many trials using 1 to 2 g daily, have been shown to be effective in prevention and treatment of depressive disorders,[63],[64],[65] with EPA having better data for efficacy than DHA.[66] In bipolar disorders, there is strong evidence that omega-3 fatty acids are helpful in depression, but not for attenuating mania.[67]
S-Adenosylmethionine (SAMe)
SAMe is produced in vivo from homocysteine and 5-MTHF, and it is also available as a supplement. It is the body’s major methyl-group donor and it is vital for membrane function and neurotransmission.[68] In divided doses totaling 800 to 1,600 mg daily, SAMe has been shown to be just as effective as tricyclic antidepressants, with a lower side effect profile.[69] It also has been shown to have a beneficial effect in depressed individuals where medication was not fully resolving their symptoms.[70] It should be used with caution in bipolar disorder, as it can trigger mania.[71]
Amino Acids
5-hydroxytryptophan (5-HTP) is the rate-limiting intermediary in the synthesis of serotonin from L-tryptophan. The dosage of 5-HTP depends on condition, ranging from 50 mg to 3 g daily in short studies.[72],[73],[74],[75] Commonly, it is dosed at 50 to 100 mg, one to three times daily, with the some of the best evidence at this dose seen in anxiety.[76],[77] For depression, clinical trials have used 400 to 900 mg per day in divided doses.[78],[79],[80] Caution is advised for those on selective serotonin reuptake inhibitors (SSRIs).
N-acetylcysteine (NAC), a derivative of L-cysteine, but more stable,[81] is well known for its function as an antioxidant and precursor to glutathione,[82] acts as a mucolytic,[83] has anti-inflammatory properties,[84] and is the treatment of choice for acetaminophen-induced hepatic necrosis.[85] At 1,000 mg, two to three times daily, this sulfhydryl molecule also possesses efficacy in numerous neuropsychiatric conditions.[86],[87],[88] It appears to increase the uptake of cysteine, which activates a reverse transport of glutamate into the extracellular space. Restoring glutamate to the extracellular space inhibits more glutamate release, thereby improving compulsive behaviors.[89]
Taurine can be synthesised in vivo from cysteine. It stabilizes cell membranes, is an osmoregulator, assists in bile acid conjugation, contributes to cardiac contractility, inhibits platelet aggregation, is an antiarrhythmic and anticonvulsant, and—last but not least—also functions as a neurotransmitter.[90],[91] While direct clinical trials of taurine on mood disorders may not exist, taurine has been shown to inhibit the release of excitatory neurons, like glutamate; act as a GABA agonist; inhibit TNFα; and increase ATP production.[92],[93]
Hormones
The use of pharmaceutical hormone replacement therapies (oestrogen, progesterone, and testosterone) will not be discussed in this article, but the prudent use of dehydroepiandrosterone (DHEA) and pregnenolone will be.
Pregnenolone is a ubiquitously produced endogenous neurosteroid, mostly made in the brain and adrenal glands from cholesterol. It is known as the master steroid hormone, since all steroid hormones, including cortisol, aldosterone, allopregnanolone, DHEA, progesterone, and testosterone, are made from it.[94] Pregnenolone is thought to interact with the cannabinoid receptor 1 (CB1), exerting antidepressant effects.[95] Pregnenolone and its metabolites have also been shown to modulate GABAA; N-methyl-D-aspartate (NMDA); and cholinergic, dopaminergic, and neurotrophic systems, thus affecting neuronal excitability.[96],[97]
In individuals with mood disorders, doses have ranged from 5 to 500 mg daily, with typical dosing of 50 to 100 mg daily. Monitoring serum pregnenolone levels every three to six months is advisable. Studies in both bipolar disorder and depression have shown significant improvements in symptoms.[98],[99]
DHEA is the most abundant neurosteroid hormone in the human body, secreted by the adrenal gland and produced in the brain.[100] As a precursor to male and female sex hormones, DHEA has been shown to be effective in many health conditions, but germane to this paper, doses of 30 to 500 mg daily have been shown to be helpful in depression and dysthymia.[101],[102] DHEA-S, the major circulating metabolite of DHEA, is not subject to day-to-day and diurnal changes that DHEA is.[103],[104] For this reason, DHEA-S should be tested prior to administering the hormone to ensure it may be of benefit, as well as monitored every three to six months. Excessive administration of DHEA can cause acne and hirsutism, and as a precursor to oestrogen and testosterone, there is a theoretical risk that long-term use could lead to hormone-sensitive cancers, especially if DHEA-S becomes elevated.
Botanicals
What doesn’t Curcuma longa (turmeric) do? Its virtues are endless, but one may not be aware of is its efficacy in depressive disorders. Studies have shown that just 1,000 mg of the herb daily is as effective as 20 mg of fluoxetine, and when used in combination with the medication, response rates for those with major depression rose from 65% to 78%.[105] A 2017 meta-analysis showed, again, its efficacy in depression.[106] It also has been shown to reduce anxiety.[107] It is postulated that turmeric inhibits the activity of both monoamine oxidase (MAO)-A and MAO-B, increases the levels of neurotrophic factors (particularly brain-derived neurotrophic factor [BDNF]), and modulates the serotonin and dopamine neurotransmission in the brain.[108]
Rhodiola rosea (rhodiola) is a wonderful plant that thrives in cold regions and high altitudes, and it is notorious for its ability to increase resistance to physical, chemical, and biological stressors.[109] In vitro and animal studies have shown the constituents rhodioloside, salidroside, and tyrosol regulate the activity of serotonin, dopamine, and norepinephrine, as well as inhibit MAO-A.[110],[111],[112] In depressed individuals, 340 mg one to two times daily has been shown to decrease overall depressive symptoms, emotional instability, insomnia, and somatisation.[113],[114] Typically, dosages range from 200 to 600 mg daily, depending on the percentage of active constituents. Caution should be used in those with bipolar disorder, who are prone to manic episodes when given antidepressants or stimulants.[115]
Crocus sativus (saffron) is a well-known, brilliant, yellow-red, and precious spice that mostly grows in Iran, Greece, Morocco, and India, and it is one of the most expensive botanicals in the world.[116] It has a long history of traditional use, and it is considered to be an antispasmodic, thymoleptic, carminative, cognition enhancer, aphrodisiac, and emmenagogue.[117] This revered spice has also been shown clinically to benefit attention-deficit/hyperactivity disorder (ADHD),[118] Alzheimer’s disease,[119],[120] anxiety,[121] and depression.[122] It is theorised that safranal (a carotenoid found in saffron) interacts with the GABAergic system,[123] modulates levels of serotonin (possibly by inhibiting reuptake),[124] and alters levels of dopamine and norepinephrine.[125] Standardized extracts containing 2% safranal, 2% crocin, and small amounts of picrocrocin (% in studies unspecified), dosed at 15 mg twice daily, have been shown to significantly reduce numerous parameters of depression.[126],[127],[128] Saffron is very safe and has no known drug-herb interactions.[129]
The aforementioned text is not an exhaustive list of safe and effective interventions to mood disorders, but rather a consolidation of what has better evidence clinically, both from published human studies and this author’s personal experience. As with any health condition, individuals should not self-treat, but rather seek out a qualified healthcare professional to discuss their health concerns and options.
References
[1] Cleveland Clinic. Mood Disorders [Internet]. Cleveland (OH): Cleveland Clinic; 2018 [cited 2019 Aug 18]. Available from: https://my.clevelandclinic.org/health/diseases/17843-mood-disorders
[2] Johns Hopkins Medicine. Mood Disorders [Internet]. Baltimore (MD): The Johns Hopkins University, The Johns Hopkins Hospital, and Johns Hopkins Health System [cited 2019 Aug 18]. Available from: https://www.hopkinsmedicine.org/health/conditions-and-diseases/mood-disorders
[3] National Institute of Mental Health. Any Mood Disorder [Internet]. Atlanta (GA): U S Department of Health and Human Services; 2017 [cited 2019 Aug 18]. Available from: https://www.nimh.nih.gov/health/statistics/any-mood-disorder.shtml
[4] World Health Organization. Mental Disorders [Internet]. Geneva, Switzerland: World Health Organization; 2018 [cited 2019 Aug 18]. Available from: https://www.who.int/news-room/fact-sheets/detail/mental-disorders
[5] Stuart L, et al. Seasonal Affective Disorder. Am Fam Physician. 2012;86(11):1037-41.
[6] World Health Organization. The global burden of disease: 2004 update. Geneva, Switzerland: World Health Organization; 2008.
[7] Gregory C. Mood Disorders [Internet]. New York: Remedy Health Media, LLC; 2019 [cited 2019 Aug 18]. Available from: https://www.psycom.net/mood-disorders/
[8] Malhi GS, Mann JJ. Depression. Lancet. 2018 Nov 24;392(10161):2299-312.
[9] Pigott HE, et al. Efficacy and effectiveness of antidepressants: current status of research. Psychother Psychosom. 2010;79(5):267-79.
[10] Pompili M, et al. Improving adherence in mood disorders: the struggle against relapse, recurrence and suicide risk. Expert Rev Neurother. 2009;9:985-1004.
[11] Mayo Clinic Staff. Depression (major depressive disorder) [Internet]. Rochester (MN): Mayo Clinic; 2019 [cited 2019 Aug 18]. Available from: https://www.mayoclinic.org/diseases-conditions/depression/symptoms-causes/syc-20356007
[12] Lau JY, Eley TC. The genetics of mood disorders. Annu Rev Clin Psychol. 2010;6:313-37.
[13] Archer T, et al. Epigenetic Modulation of Mood Disorders. J Genet Syndr Gene Ther. 2013 Feb 11; 4(120):1000120.
[14] Detera-Wadleigh SD, McMahon FJ. Genetic association studies in mood disorders: issues and promise. Int Rev Psychiatry. 2004 Nov;16(4):301-10.
[15] Lynch B. Dirty Genes. New York: Harper One; 2018.
[16] Amare AT, et al. Pharmacogenomics in the treatment of mood disorders: Strategies and Opportunities for personalized psychiatry. EPMA J. 2017 Sep 5;8(3):211-27.
[17] Brennan FX, et al. A Naturalistic Study of the Effectiveness of Pharmacogenetic Testing to Guide Treatment in Psychiatric Patients With Mood and Anxiety Disorders. Prim Care Companion CNS Disord. 2015;17(2):10.4088/PCC.14m01717.
[18] Pickar D, Rubinow K. Pharmacogenomics of psychiatric disorders. Trends Pharmacol Sci. 2001 Feb;22(2):75-83.
[19] Qureshi NA, Al-Bedeh AM. Mood disorders and complementary and alternative medicine: a literature review. Neuropsychiatr Dis Treat. 2013;9:639-58.
[20] Elkins G, et al. Complementary and alternative medicine use by psychiatric inpatients. Psychol Rep. 2005;96:163-6.
[21] Simon GE, et al. Mental health visits to complementary and alternative medicine providers. Gen Hosp Psychiatry. 2004;26:171-7.
[22] Davison KM, Kaplan BJ. Nutrient intakes are correlated with overall psychiatric functioning in adults with mood disorders. Can J Psychiatry. 2012 Feb;57(2):85-92.
[23] Byrne A, Byrne DG. The effect of exercise on depression, anxiety and other mood states: a review. J Psychosom Res. 1993 Sep;37(6):565-74.
[24] Lakhan SE, Vieira KF. Nutritional therapies for mental disorders. Nutr J. 2008;7:2.
[25] Foster JA, McVey Neufeld KA. Gut-brain axis: how the microbiome influences anxiety and depression. Trends Neurosci. 2013 May;36(5):305-12.
[26] Sherwin E, at al. A gut (microbiome) feeling about the brain. Curr Opin Gastroenterol. 2016 Mar;32(2):96-102.
[27] Gaby A. Nutritional Medicine. 2nd ed. Concord (NH): Fritz Perlberg Publishing; 2017.
[28] Hutto BR. Folate and cobalamin in psychiatric illness. Compr Psychiatry. 1997;38(6):305-14
[29] Hector M, Burton JR. What are the psychiatric manifestations of vitamin B12 deficiency? J Am Geriatr Soc. 1988;36:1105-12.
[30] Coppen A, Bolander-Gouaille C. Treatment of depression: time to consider folic acid and vitamin B12. J Psychopharmacol. 2005 Jan;19(1):59-65.
[31] Reading CM. X-linked dominant manic-depressive illness: linkage with Xg blood group, red-green color blindness, and vitamin B12 deficiency. J Orthomolecular Psychiatry. 1979;8:68-77.
[32] Van Tiggelen CJM, et al. Assessment of vitamin B12 status in CSF. Am J Psychiatry. 1984;141:136-7.
[33] McNulty H, et al. Folate [Internet]. Corvallis (OR): Micronutrient Information Center, Linus Pauling Institute, Oregon State University; 2014 [cited 2019 Aug 18]. Available from: https://lpi.oregonstate.edu/mic/vitamins/folate
[34] Alpert JE, Fava M. Nutrition and depression: the role of folate. Nutr Rev. 1997 May;55(5):145-9.
[35] Coppen AA, Bailey J. Enhancement of the antidepressant action of fluoxetine by folic acid: a randomised, placebo controlled trial. J Affect Dis. 2000;60:121-31.
[36] Behzadi, AH, et al. Folic acid efficacy as an alternative drug added to sodium valproate in the treatment of acute phase of mania in bipolar disorder: a double-blind randomized controlled trial. Acta Psychiatr Scand. 2009;120(6):441-5.
[37] Pietrzik K, et al. Folic acid and L-5-methyltetrahydrofolate: comparison of clinical pharmacokinetics and pharmacodynamics. Clin Pharmacokinet. 2010 Aug;49(8):535-48.
[38] Scaglione F, Panzavolta G. Folate, folic acid and 5-methyltetrahydrofolate are not the same thing. Xenobiotica. 2014 May;44(5):480-8.
[39] Bender DA. Vitamin B6 requirements and recommendations. Eur J Clin Nutr. 1989;43:289-309.
[40] Clayton PT. B6-responsive disorders: a model of vitamin dependency. J Inherit Metab Dis. 2006;29(2-3):317-26.
[41] Miller JW, et al. Effect of vitamin B-6 deficiency on fasting plasma homocysteine concentrations. Am J Clin Nutr. 1992 Jun;55(6):1154-60.
[42] Berger, et al. Dose response, coasting, and differential fiber vulnerability in human toxic neuropathy: a prospective study of pyridoxine neurotoxicity. Neurology. 1992;42:1367-70.
[43] Krishnan AV, Feldman D. Mechanisms of the anti-cancer and anti-inflammatory actions of vitamin D. Annu Rev Pharmacol Toxicol. 2011;51:311-36.
[44] Moyad MA. Vitamin D: a rapid review. Urol Nurs. 2008 Oct;28(5):343-9.
[45] Autier P, et al. Vitamin D status and ill health: a systematic review. Lancet Diabetes Endocrinol. 2014 Jan;2(1):76-89.
[46] Spedding S. Vitamin D and depression: a systematic review and meta-analysis comparing studies with and without biological flaws. Nutrients. 2014 Apr 11;6(4):1501-18.
[47] Gloth FM 3rd, et al. Vitamin D vs broad spectrum phototherapy in the treatment of seasonal affective disorder. J Nutr Health Aging. 1999;3(1):5-7.
[48] Carrol A. Why Take Vitamin D Supplements if They Don’t Improve Health? [Internet]. Chicago: JAMA Forum; 2016 [cited 2019 Aug 18]. Available from: https://newsatjama.jama.com/2016/03/24/jama-forum-why-take-vitamin-d-supplements-if-they-dont-improve-health/
[49] Szabo L. The Man Who Sold America On Vitamin D –And Profited in the Process. Medscape. 2018 Aug 24.
[50] Wallace TC, et al. Multivitamin/mineral supplement contribution to micronutrient intakes in the United States, 2007-2010. J Am Coll Nutr. 2014;33(2):94-102.
[51] DiNicolantonio JJ, et al. Subclinical magnesium deficiency: a principal driver of cardiovascular disease and a public health crisis. Open Heart. 2018;5(1):e000668.
[52] Altura B. Basic biochemistry and physiology of magnesium: a brief review. Magnes Trace Elem. 1991-2;10:167-71.
[53] Wester PO. Magnesium. Am J Clin Nutr. 1987;45:1305-12.
[54] Imada Y, et al. Relationships between serum magnesium levels and clinical background factors in patients with mood disorders. Psychiatry Clin Neurosci. 2002 Oct;56(5):509-14.
[55] Tarleton EK, et al. Role of magnesium supplementation in the treatment of depression: A randomized clinical trial. PLoS One. 2017 Jun 27;12(6):e0180067.
[56] Serefko A, et al. Magnesium and depression. Magnes Res. 2016 Mar 1;29(3):112-9.
[57] McCall KA, et al. Function and mechanism of zinc metalloenzymes. J Nutr. 2000 May;130(5S Suppl):1437S-46S.
[58] Nowak G, et al. Effect of zinc supplementation on antidepressant therapy in unipolar depression: a preliminary placebo-controlled study. Pol J Pharmacol. 2003;55(6):1143-7.
[59] Li Z et al. Dietary zinc and iron intake and risk of depression: A meta-analysis. Psychiatry Res. 2017;251:41-7.
[60] Institute of Medicine (US) Panel on Micronutrients. Dietary reference intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. Washington (DC): National Academies Press; 2001:442-501.
[61] Maroon JC, Bost JW. Omega-3 fatty acids (fish oil) as an anti-inflammatory: an alternative to nonsteroidal anti-inflammatory drugs for discogenic pain. Surg Neurol. 2006 Apr;65(4):326-31.
[62] Kang JX, Weylandt KH. Modulation of inflammatory cytokines by omega-3 fatty acids. Subcell Biochem. 2008;49:133-43.
[63] Su KP, et al. Omega-3 fatty acids in the prevention of interferon-alpha-induced depression: results from a randomized, controlled trial. Biol Psychiatry. 2014 Oct 1;76(7):559-66.
[64] Grosso G, et al. Role of omega-3 fatty acids in the treatment of depressive disorders: a comprehensive meta-analysis of randomized clinical trials. PLoS One. 2014 May 7;9(5):e96905.
[65] Lin PY, Su KP. A meta-analytic review of double-blind, placebo-controlled trials of antidepressant efficacy of omega-3 fatty acids. J Clin Psychiatry. 2007 Jul;68(7):1056-61.
[66] Martins JG. EPA but not DHA appears to be responsible for the efficacy of omega-3 long chain polyunsaturated fatty acid supplementation in depression: evidence from a meta-analysis of randomized controlled trials. J Am Coll Nutr. 2009 Oct;28(5):525-42.
[67] Sarris J, et al. Omega-3 for bipolar disorder: meta-analyses of use in mania and bipolar depression. J Clin Psychiatry. 2012 Jan;73(1):81-6.
[68] Carney MW, et al. S-Adenosylmethionine and affective disorder. Am J Med. 1987;83(Suppl 5A):104-6.
[69] Sharma A, et al. S-Adenosylmethionine (SAMe) for Neuropsychiatric Disorders: A Clinician-Oriented Review of Research. J Clin Psychiatry. 2017 Jun;78(6):e656-67.
[70] Papakostas GI, et al. S-adenosyl methionine (SAMe) augmentation of serotonin reuptake inhibitors for antidepressant nonresponders with major depressive disorder: a double-blind, randomized clinical trial. Am J Psychiatry. 2010 Aug;167(8):942-8.
[71] Carney MW, et al. The switch mechanism and the bipolar/unipolar dichotomy. Br J Psychiatry. 1989 Jan;154:48-51.
[72] Kahn RS, Westenberg HG. L-5-hydroxytryptophan in the treatment of anxiety disorders. J Affect Disord. 1985;8:197-200.
[73] Sarzi Puttini P, Caruso I. Primary fibromyalgia syndrome and 5-hydroxy-L-tryptophan: a 90-day open study. J Int Med Res. 1992;20:182-9.
[74] Angst J, et al. The treatment of depression with L-5-hydroxytryptophan versus imipramine. Results of two open and one double-blind study. Arch Psychiatr Nervenkr. 1977;224:175-86.
[75] van Praag HM, et al. A pilot study of the predictive value of the probenecid test in application of 5-hydroxytryptophan as antidepressant. Psychopharmacologia. 1972;25:14-21.
[76] Kahn RS, Westenberg HG. L-5-hydroxytryptophan in the treatment of anxiety disorders. J Affect Disord. 1985;8:197-200.
[77] Kahn RS, et al. Effect of a serotonin precursor and uptake inhibitor in anxiety disorders; a double-blind comparison of 5-hydroxytryptophan, clomipramine and placebo. Int Clin Psychopharmacol. 1987;21:33-45.
[78] Nakajima T, et al. Clinical evaluation of 5-hydroxy-L-tryptophan as an antidepressant drug. Folia Psychiatr Neurol Jpn. 1978;32:223-30.
[79] Poldinger W, et al. A functional-dimensional approach to depression: serotonin deficiency as a target syndrome in a comparison of 5-hydroxytryptophan and fluvoxamine. Psychopathology. 1991;24:53-81.
[80] Jangid P, et al. Comparative study of efficacy of l-5-hydroxytryptophan and fluoxetine in patients presenting with first depressive episode. Asian J Psychiatr. 2013 Feb;6(1):29-34.
81 Ziment I. Acetylcysteine: a drug with an interesting past and a fascination future. Respiration. 1986;50(Suppl 1):26-30.
[82] James SJ, et al. Thimerosal neurotoxicity is associated with glutathione depletion: protection with glutathione precursors. Neurotoxicology. 2005 Jan;26(1):1-8.
[83] Sadowska A, et al. Role of N-acetylcysteine in the management of COPD. Int J Chron Obstruct Pulmon Dis. 2006 Dec;1(4):425-34.
[84] Palacio JR, et al. Anti-inflammatory properties of N-acetylcysteine on lipopolysaccharide-activated macrophages. Inflamm Res. 2011 Jul;60(7):695-704.
[85] Harrison PM, et al. Improvement by acetylcysteine of hemodynamics and oxygen transport in fulminant hepatic failure. N Engl J Med. 1991 Jun 27;324(26):1852-7.
[86] Ooi SL, et al. N-Acetylcysteine for the Treatment of Psychiatric Disorders: A Review of Current Evidence. Biomed Res Int. 2018;2018:2469486.
[87] Dean O, et al. N-acetylcysteine in psychiatry: current therapeutic evidence and potential mechanisms of action. J Psychiatry Neurosci. 2011;36(2):78-86.
[88] Minarini A, et al. N-acetylcysteine in the treatment of psychiatric disorders: current status and future prospects. Expert Opin Drug Metab Toxicol. 2017 Mar;13(3):279-92.
[89] Grant JE, et al. N-acetylcysteine, a glutamate modulator, in the treatment of trichotillomania: a double-blind, placebo-controlled study. Arch Gen Psychiatry. 2009;66:756-63.
[90] Hayces KC. A review on the biological function of taurine. Nutr Rev. 1976;34:161-5.
[91] Ripps H, Shen W. Review: taurine: a “very essential” amino acid. Mol Vis. 2012;18:2673-86.
[92] Islambulchilar M, et al. Taurine attenuates chemotherapy-induced nausea and vomiting in acute lymphoblastic leukemia. Amino Acids. 2015 Jan;47(1):101-9.
[93] Ripps H, Shen W. Review: taurine: a “very essential” amino acid. Mol Vis. 2012;18:2673-86.
[94] Devlin TM, ed. Textbook of Biochemistry with Clinical Correlations. 3rd ed. New York: Wiley-Liss Inc.; 1992.
[95] Brown ES, et al. A randomized, double-blind, placebo-controlled trial of pregnenolone for bipolar depression. Neuropsychopharmacology. 2014;39(12):2867-73.
[96] Ritsner MS, et al. Pregnenolone and dehydroepiandrosterone as an adjunctive treatment in schizophrenia and schizoaffective disorder: an 8-week, double-blind, randomized, controlled, 2-center, parallel-group trial. J Clin Psychiatry. 2010;71(10):1351-62.
[97] Marx CE, et al. Proof-of-concept trial with the neurosteroid pregnenolone targeting cognitive and negative symptoms in schizophrenia. Neuropsychopharmacology. 2009;34(8):1885-903.
[98] Osuji IJ, et al. Pregnenolone for cognition and mood in dual diagnosis patients. Psychiatry Res. 2010 Jul 30;178(2):309-12.
[99] George MS, et al. CSF neuroactive steroids in affective disorders: pregnenolone, progesterone, and DBI. Biol Psych. 1994;35:775-80.
[100] Kroboth PD, et al. DHEA and DHEA-S: A review. J Clin Pharmacol. 1999;39:327-48.
[101] Wolkowitz OM, et al. Double-blind treatment of major depression with dehydroepiandrosterone. Am J Psychiatry. 1999;156:646-9.
[102] Bloch M, et al. Dehydroepiandrosterone treatment of midlife dysthymia. Biol Psychiatry. 1999;45:1533-41.
[103] Kamin HS, Kertes DA. Cortisol and DHEA in Development and Psychopathology. Horm Behav. 2017 Mar;89:69-85.
[104] Starka L, et al. Dehydroepiandrosterone: a neuroactive steroid. J Steroid Biochem Mol Biol. 2015;145:254-60.
[105] Sanmukhani J, et al. Efficacy and safety of curcumin in major depressive disorder: a randomized controlled trial. Phytother Res. 2014;28(4):579-85.
[106] Nq QX, et al. Clinical Use of Curcumin in Depression: A Meta-Analysis. J Am Med Dir Assoc. 2017 Jun 1;18(6):503-8.
[107] Esmaily H, et al. An investigation of the effects of curcumin on anxiety and depression in obese individuals: A randomized controlled trial. Chin J Integr Med. 2015 May;21(5):332-8.
[108] Kulkarni SK, Dhir A. An Overview of Curcumin in Neurological Disorders. Indian J Pharm Sci. 2010 Mar-Apr; 72(2):149-54.
[109] Kelly GS. Rhodiola rosea: a possible plant adaptogen. Altern Med Rev. 2001;6:293-302.
[110] Chen Q, et al. The effects of Rhodiola rosea extract on 5-HT level, cell proliferation and quantity of neurons at cerebral hippocampus of depressive rats. Phytomedicine. 2009;16(9):830-8.
[111] Panossian A, et al. Comparative study of Rhodiola preparations on behavioral despair of rats. Phytomedicine. 2008;15(1-2):84-91.
[112] van Diermen D, et al. Monoamine oxidase inhibition by Rhodiola rosea L. roots. J Ethnopharmacol. 2009;122(2):397-401.
[113] Darbinyan G, et al. Clinical trial of Rhodiola rosea L. extract SHR-5 in the treatment of mild to moderate depression. Nord J Psychiatry. 2007;61:343-8.
[114] Ross M. Rhodiola rosea (SHR-5), Part 2: A standardized extract of Rhodiola rosea is shown to be effective in the treatment of mild to moderate depression. Holist Nurs Pract. 2014 May-Jun;28(3):217-21.
[115] Brown RP, et al. Rhodiola rosea: A Phytomedicinal Overview. HerbalGram. 2002;56:40-52.
[116] Baraghani A. What Is Saffron, the World’s Most Legendary Spice? [Internet]. New York: Healthyish, Bon Appetit; 2018 [cited 2019 Aug 18]. Available from: https://www.bonappetit.com/story/what-is-saffron
[117] Rios JL, et al. An update review of saffron and its active constituents. Phytother Res. 1996;10:189-93.
[118] Baziar S, et al. Crocus sativus L. Versus Methylphenidate in Treatment of Children with Attention-Deficit/Hyperactivity Disorder: A Randomized, Double-Blind Pilot Study. J Child Adolesc Psychopharmacol. 2019 Apr;29(3):205-12.
[119] Akhondzadeh S, et al. Saffron in the treatment of patients with mild to moderate Alzheimer’s disease: a 16-week, randomized and placebo-controlled trial. J Clin Pharm Ther. 2010 Oct;35(5):581-8.
[120] Akhondzadeh S, et al. A 22-week, multicenter, randomized, double-blind controlled trial of Crocus sativus in the treatment of mild-to-moderate Alzheimer’s disease. Psychopharmacology. 2010;207:637-43.
[121] Mazidi M, et al. A double-blind, randomized and placebo-controlled trial of Saffron (Crocus sativus L.) in the treatment of anxiety and depression. J Complement Integr Med. 2016 Jun 1;13(2):195-9.
[122] Dwyer AV, et al. Herbal Medicines, other than St. John’s Wort, in the Treatment of Depression: A Systematic Review. Altern Med Rev. 2011 Mar;16(1):40-9.
[123] Marañón JA, et al. GABA receptors mediates the activity of safranal from IRIDAFRAN saffron extract. Int Soc Nutraceutical Func Foods. 2012. Poster presentation.
[124] Mazidi M, et al. A double-blind, randomized and placebo-controlled trial of Saffron (Crocus sativus L.) in the treatment of anxiety and depression. J Complement Integr Med. 2016 Jun 1;13(2):195-9.
[125] Talaei A, et al. Crocin, the main active saffron constituent, as an adjunctive treatment in major depressive disorder: a randomized, double-blind, placebo-controlled, pilot clinical trial. J Affect Disord. 2015 Mar 15;174:51-6.
[126] Akhondzadeh S, et al. Crocus sativus L. in the Treatment of Mild to Moderate Depression: A Double-blind, Randomized and Placebo-controlled Trial. Phytother Res. 2005 Feb;19(2):148-51.
[127] Noorbala AA, et al. Hydro-alcoholic extract of Crocus sativus L. versus fluoxetine in the treatment of mild to moderate depression: a double-blind, randomized pilot trial. J Ethnopharmacol. 2005 Feb 28;97(2):281-4.
[128] Akhondzadeh S, et al. Comparison of Crocus sativus L. and imipramine in the treatment of mild to moderate depression: A pilot double-blind randomized trial. BMC Complement Altern Med. 2004 Sep 2;4:12.
[129] Stargrove MB, et al. Herb, Nutrient and Drug Interactions. Maryland Heights (MO): Mosby; 2008.





