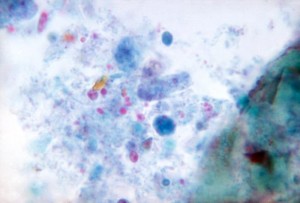
First, some background to this parasite and a brief discussion of what may be used pharmaceutically and then a review of how a non-drug approach may be applied through the understanding of the mechanisms employed by the organism and its relative susceptibility to eradication strategies.
Dientamoeba fragilis (D. fragilis)is a trichomonad (a genus of anaerobic protists that are parasites of vertebrates) parasite found in the gastrointestinal tract of humans and implicated as a cause of gastrointestinal disease. D. fragilis has been found in most parts of the world in both rural and cosmopolitan areas. Infection with D. fragilis is called Dientamoebiasis and is associated variously with symptoms of abdominal pain, diarrhoea, weight loss, and fever.[1]
Dientamoeba fragilis is a parasite that causes gastrointestinal problems. Despite its name, Dientamoeba fragilis is not an amoeba but a flagellate. This protozoan parasite produces trophozoites; cysts have not been identified. Infection may be either symptomatic or asymptomatic.
Under Appreciated?
Ever since its first description in 1918, D. fragilis has struggled to gain recognition as a significant pathogen. There is little justification for this neglect, however, since there exists a growing body of case reports from numerous countries around the world that have linked this protozoal parasite to numerous clinical manifestations. A number of studies have even incriminated D. fragilis as a cause of irritable bowel syndrome,[2] allergic colitis,[3] and diarrhoea in human immunodeficiency virus patients. As it is often found in people with other co infections there has been some on-going doubt about its validity as a pathogen.
In part this is reinforced by the fact colonisation may occur without development of disease. In adults, asymptomatic colonisation is present in 75-85% of individuals affected by the parasite. In children, the opposite is true; disease develops in as many as 90% of those colonised.
However, numerous reports from many different parts of the world continued to substantiate the association of D. fragilis with clinical symptoms.[4]
Symptoms
Adults may experience one or more of the following, in children, pain varies with regards to location, duration, and character.
Other GI complaints include the following:
- Anorexia
- Weight loss
- Nausea
- Vomiting
- Bloating
- Flatulence
- Alternating constipation and diarrhoea
Non-intestinal complaints include the following:
- Headache
- Fever
- Malaise
- Fatigue
- Irritability
- Weakness
- Pruritus
- Urticaria
Frequency
A North American study in 2006, stated: “The prevalence of D. fragilis would surprise most clinicians. A review of stool-examination reports at the Cadham Provincial Laboratory in Winnipeg, which handles 80%–90% of stool examinations for parasites done in Manitoba, revealed that the incidence of this parasite was second only to Blastocystis hominis and far in excess of more commonly incriminated parasites such as Giardia lamblia/intestinalis, Entamoeba histolytica/dispar and Cryptosporidium parvum. Coinfection with B. hominis and D. fragilis was also common.” [5]
A high prevalence of D.fragilis has been reported in faecal samples collected from patients attending complementary medicine practitioners in the UK. Specimens were collected directly after passing into sodium acetate-acetic acid-formalin (SAF).
During two observation periods in 2002-04 and 2005-07, a D. fragilis prevalence of 14.6% (n = 543) and 16.9% (n = 421), respectively, was recorded. These results confirm a surprisingly high prevalence of D. fragilis among a selected population.[6]
D. fragilis infections as mentioned above are often associated with other intestinal parasites. Ayadi and Barri investigated 1,497 confirmed D. fragilis cases and found coinfections with
- E. vermicularis in only 5% but with
- B. hominis in 40.3% of the cases and with
- Endolimax nana, Entamoeba coli, and Giardia intestinalis in 24, 6, and 5.7%
..of the cases; respectively. This high coincidence of infection with other organisms that are transmitted via the faecal-oral route suggests the possibility of a similar mode of transmission for D. fragilis.[7] In children particularly consider the co-existance of pin worms (E. vermicularis), these can checked for using the sellotape test:
Place sellotape over the anus of the child before retiring to bed and in the morning see if worms have become attached. These will need treatment as well
Medical Treatment
Drug Therapy includes:
- Diphetarsone
- Tetracycline
- Carbarsone
- Metronidazole
- Iodoquinol
- Erythromycin
- Hydroxychinoline
- Paromomycin and
- Secnidazole
- Humatin
Mechanism of action varies with the drug class but may include the following:
- Inhibition of microtubules, causing irreversible block of glucose uptake
- Tubulin polymerisation inhibition
- Depolarising neuromuscular blockade
- Cholinesterase inhibition
- Increased cell membrane permeability, resulting in intracellular calcium loss
- Vacuolisation of the schistosome tegument
- Increased cell membrane permeability to chloride ions via chloride channels alteration
Though there remains some questions about whether these therapies directly target the pathogen or disrupt the bacterial colonies supporting its existence. This pathogen appears only, as with most, to be able to colonise a GI tract to the point of being symptomatic by co-existing with dysbiotic changes to the microbiota.
However, there is no general consensus on what is best practice in treating D. fragilis infections. There are also no data available on the mechanisms of immunity for this parasite.
Risks of Medications
Side effects have also been reported for other therapeutic agents used to treat D. fragilis infections. Transient liver function abnormalities were observed in several patients treated with Diphetarsone . Tetracycline has limited usefulness in children because of its well-established deleterious effect on dental development. Presently, Iodoquinol and Tetracycline are the most commonly employed medications, but a recent study found the antiaomebic drug Secnidazole to be highly effective. D. fragilis was eradicated in 34 of 35 patients after receiving a single dose of Secnidazole. A second dose was required only for one patient.[8],[9]
Natural Therapy
The successful eradication of D.fragilis will not occur with either therapy on a consistent basis, that is drugs and natural therapies have a high failure rate. In part this is due to the poor understanding of the organisms requirements for existence and partly because it seems to develop a very good tolerance to medicines and natural agents.
This resistance to eradication may be a reflection of the altered microbial communities as suggested by frequent co-infection and a broad spectrum of intervention/eradication strategies as well as a nutritive strategy and bacterial re-colonisation support may not only improve eradication but also reduce post treatment complications.
In children the need for successful management of this organism is just as important but requires care in the selection of medicines and or non drug treatment.
Regardless of the eradication therapy chosen – this first intervention recommendation is suggested as a suitable method of managing ecology needs in the small and principle place of residence – the large intestine.
Consider
- Saccharomyces Boulardii up to 30 billion CFU’s daily
- Bifidus (Colonic Bacteria)species and lower small intestinal species in divided doses with food up to 30 billion CFU’s
Consider these options for eradication
- Standardised Extract of Garlic 1000mg – 2000mg per day – this is a low risk broad antifungal/parasitic suggestion and may be used away from the probiotics suggested above.
- A combination of Wormwood, Berberine, Citrus Seed Extract & Black Walnut prepared in active forms. These traditional herbs require liver enzyme monitoring if used for more than 30 consecutive days.
- Colloidal Silver – the use of silver solutions are indicated but there is no direct evidence of efficacy in this parasite treatment. However it is easy to administer and if inclined to consider this: Silver 50 once per day
- Artemesia standardised extract
Control measures to limit spread of parasites include the following:
- Disinfect surfaces and equipment handled by children infected by the parasite.
- Disinfect nappy areas.
- Separate nappy areas from food preparation sites.
- Educate about handwashing techniques.
- Improve personal hygiene.
- Separate symptomatic individuals.
Comment
Due to the high frequency of coinfection with B.hominis, a joint strategy for the treatment of both parasites along with suitable innate defence support is recommended. I have written about B.hominis in a previous post.
References
[1] Johnson, E. H., J. J. Windsor, and G. C. Clark. 2004. Emerging from obscurity: biological, clinical, and diagnostic aspects of Dientamoeba fragilis.Clin. Microbiol. Rev. 17:553-570 View Abstract
[2] Yakoob J, Jafri W, Beg MA, Abbas Z, Naz S, Islam M, Khan R. Blastocystis hominis and Dientamoeba fragilis in patients fulfilling irritable bowel syndrome criteria. Parasitol Res. 2010 Aug;107(3):679-84. Epub 2010 Jun 8 View Abstract
[3] Yamamoto-Furusho JK, Torijano-Carrera E. Intestinal protozoa infections among patients with ulcerative colitis: prevalence and impact on clinical disease course. Digestion. 2010;82(1):18-23. Epub 2010 Feb 9. View Abstract
[4] Addadi, K., Y. Le Corroller, Y. Guy, and A. Tabet-Derraz. 1972. Sur le role pathogene possible de Dientamoeba fragilis. Bull. Soc. Pathol. Exot. Fil. 65:274-275. View Abstract
[5] Lagacé-Wiens PR, VanCaeseele PG, Koschik C. “Dientamoeba fragilis: an emerging role in intestinal disease”. CMAJ 175 (5): 468–9, August 2006. View Abstract
[6] Schuster H, Jackson RS. Prevalence of Dientamoeba fragilis among patients consulting complementary medicine practitioners in the British Isles. J Clin Pathol. 2009 Feb;62(2):182-4. Epub 2008 Oct 24. View Abstract
[7] Ayadi, A., and I. Bahri. 1999. Dientamoeba fragilis: flagelle pathogene. Bull. Soc. Pathol. Exot. 5:299-301. View Abstract
[8] Keystone, J. S., E. Proctor, C. Glenn, and L. Mcintyre. 1983. Safety and efficacy of diphetarsone in the treatment of amoebiasis, non-pathogenic amoebiasis and trichuriasis. Trans. R. Soc. Trop. Med. Hyg. 77:84-86. View Abstract
[9] Girginkardesler, N., S. Coskun, I. Cuneyt Balcioglu, P. Ertan, and U. Z. Ok. 2003. Dientamoeba fragilis, a neglected cause of diarrhea, successfully treated with secnidazole. Clin. Microbiol. Infect. 9:110-113. View Abstract





4 Comments. Leave new
I have this parasite – my doctor has perscribed a combination drug regimen of 750mg Metronidazole 3 times per day, 500mg Clarithromycin 2 times per day, and 650 mg Diodoquin 3 times per day – is this going to kill the parasite – the regimen is extremely nauseating on the stomach – would colloidal silver in addition to this regimen help and if so in what doseage.
Hi Michael, Great article. I have had d.frag show up in my stool twice now in the last 5 years. I did flagyl the first treatment and did my own natural protocol afterwards to repair the gut. I never fully got better but was 80 % better until a few years went by and it was getting chronically worse again. Treated again with doxy and idoquinol and was a bit better until the last few months again. Waiting on lab results to see if I have it again. I was also told to start thyroid medicine (synthroid) as my TSH has been hovering around 4.5 or higher and my FT4 is at the bottom of the normal range. My doctor doesn’t believe me but as I started the synthroid things got worse with bloating and water weight gain which led me to stop and think that I was still infected with the parasite. Could the parasite interfere with thyroid function or should I be treating this as well as I treat the parasite if in fact my test results are positive? I am thinking if my large intestine is so bloated and having issues all the time it can’t really absorb any medicines anyways or is it vice versus and more of the thyroid issue being the root cause? Thanks for any advice or information you can give me.
Hello Michael.
First of all, thank you for this sophisticated and important article.
However, from reading this article it was not so clear for me whether the using of •Metronidazole antibiotic as mentioned is effective enough to eradicate the D. fragilies? moreover, I had suffered from this parasite in my body and after taking this antibiotic it was found (by giving fecal samples once again) that it was disapeared. it seems weird since the symptoms associated with such parasite still bother my bowels. Can you please clarify more what could be the real reason for this inconvenient situation?
Hi Michael I found this very interesting article you wrote some years ago whilst looking into this protozoa. The client in question is not terribly unwell but her young daughter (age 4) is suffering with a myriad of symptoms including being very underweight and I wondered if she could potentially have this protozoa as well (she has had many GP and consultant tests and nothing has been found). It seems from your article they can be transmitted easily via stools so I am assuming it’s totally possible she picked it up from her daughter. Interestingly her stool sample also showed worms – trichuris trichiura. Shoudl I suggest she runs a stool test for her daughter? Many thanks, Julie.