 A trial to see if the ingestion of a probiotic bacterium enriched drink might have a beneficial impact on central obesity was funded by Snow Brand Milk Products company in Japan and the results were published in the European Journal of Clinical Nutrition this June 2010. [i]
A trial to see if the ingestion of a probiotic bacterium enriched drink might have a beneficial impact on central obesity was funded by Snow Brand Milk Products company in Japan and the results were published in the European Journal of Clinical Nutrition this June 2010. [i]
Whilst it may seem a stretch that bacteria can influence our body mass, (I have written a previous review) it is an area of growing interest and investigation as bacteria have previously been implicated in the metabolic storage of fat. Studies in mice have shown up to 30% greater fat storage in mice with gastrointestinal colonies of commensals rather than their skinnier counterparts operating with sterile guts.
One proposal for this is that certain bacteria (Bacteroides Thetaiotaomicron is one likely contender) are able to manipulate energy to be stored in adipocytes through a pathway that involves microbial regulation of the intestinal epithelial expression of fasting-induced adipocyte protein (Fiaf), a circulating inhibitor of lipoprotein lipase (LPL).[ii]
The microbiota can then, based on this and other studies be viewed as a metabolic “organ” exquisitely tuned to our physiology and performing functions that we have not had to evolve on our own.
Mice that are genetically obese (ob/ob) have 50% fewer Bacteroidetes, and correspondingly more Firmicutes, than their lean (+/+) siblings gut microbiota in these ob/ob mice are more effective at releasing calories from food during digestion than are the +/+ microbiota: this trait is transmissible to germ-free recipients, resulting in greater adiposity.
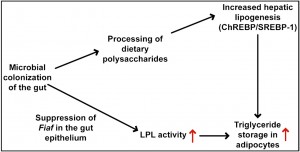
The B.Thetaiotaomicron species, a prominent mutualist in the distal intestinal habitat of adult humans likes carbohydrates as a fuel source and has a prodigious capacity for digesting otherwise indigestible dietary polysaccharides, proliferating when this is abundant.
It is possible that the bacteria in our gastrointestinal tract have developed strategies for making us – their two legged recreational vehicle – adjust to seasonal diet variations in traditional cultures. By this, they would use the periods of the year when carbohydrates from roots, fruits and other starches were dominant to provide adequate fat stores for winter seasons of limited food supply. This mechanism to provide long term homes for their progeny had a valid survival benefit for our ancestors, but the abundant intake of carbohydrates in our diets now may have persuaded some bacterial species favoured by the high sugar diets of today to continue the mechanism in anticipation of a seasonally altered diet that never arrives?
One group has estimated that individuals on a “British Diet” must ferment 50 to 65 g of hexose sugars daily to obtain the energy required to replace the 15 to 20 g (dry weight) of bacteria they excrete per day.[iii] Indicating that significant metabolic adjustments are made based on transit and food composition. Sugar consumption above this level may not be matched by normal shedding of bacterial colonies.
The idea that individual variations in metabolic efficiencies may be a significant variable in the energy balance equation is supported by several observations including variations in composition of microbiomes and that small, chronic variations in the difference between energy intake and expenditure can lead to major changes in body composition.[iv]
Food labelling implies that the caloric density of food items is a fixed value. Might it be reasonable to consider that caloric value actually varies between individual “consumers” according to the composition and operation (e.g., transit time) of their intestinal metabolic partners, and that the microbiota influences their energy balance. This would mean that those consumers with high efficiency microbes would promote energy storage (obesity); whereas lower efficiency microbes would promote leanness (efficiency is defined in this case as the energy-harvesting and storage-promoting potential of an individual’s microbiota relative to the ingested diet).
Whilst the concept is enticing the translation to humans is in its early stages of elucidation and although companies such as Nestle and Snow Brand are investing heavily results have been variable.
This paper for example, in looking at just 12 obese humans, who followed either a fat restricted or carbohydrate restricted programme were identified through bacterial analysis to have fewer Bacteroidetes (P<0.001) and more Firmicutes (P=0.002) than did the lean controls. Over time, in this case 3 months, whilst reducing their total body mass the relative abundance of Bacteroidetes increased (P<0.001) and the abundance of Firmicutes decreased (P=0.002), irrespective of diet type.[v] This implies a correlation between bacterial composition and body mass, but which comes first?
| Body Weight | BMI | Visceral Fat | |
| Pre Trial | undeclared | 24.2-30.7kg/m2 | 81.2-178.5cm2 |
| Post Trial | 1.4%
-1.1 (-1.5-0.7) kg |
1.5%
-0.4 (0.5-0.2)kg/m2 |
4.6%
-5.8 (-10.0-1.7)cm2 |
| Waist | HIP | ||
| Pre Trial | undeclared | undeclared | |
| Post Trial | 1.8%
-1.7cm (-2.1-1.4)cm |
1.5%
-1.5 (1.8-1.1)cm |
Although the root cause of obesity is excess caloric intake compared with expenditure, differences in gut microbial ecology between humans may be an important factor affecting energy homeostasis; i.e., individuals predisposed to obesity may have gut microbial communities that promote more efficient extraction and/or storage of energy from a given diet, compared with these communities in lean individuals.
So what did the Snow Brand trial reveal and what is the relationship between the science described above and the human experiment using a lactic acid bacteria.
This experiment was conducted using a multicentre, double-blind, randomised, placebo-controlled intervention trial. Subjects (n=87). Lactobacillus gasseri SBT2055 (LG2055), previously classified as L. acidophilus, is considered as a major species of human flora. L. gasseri SBT2055 is one of the strains that was isolated at the Technical Research Institute of Snow Brand Milk Products Co., Ltd (Kawagoe, Japan) from human infant or adult faeces.[vi]
This particular strain has been shown to exert a beneficial effect on rats by lowering adipocyte size and the level of leptin in specially bred rats.[vii] This suggests an additional mechanism may be involved via microbial manipulation that the earlier described Fiaf response, or it may be the same route just analysed differently. Adiponectin a known modifier of body mass increased equivalently in both groups and so is not likely to be the cause of the body changes.
In another recent paper a further lactic acid bacteria L.Plantarum (14) was used to alter body mass. Here the researchers suggest there is a further explanation saying: In conclusion, we propose that LP14 may exert a beneficial effect on the onset of diet-induced obesity by reducing the cell size of white adipose tissues, and it seems unlikely that previously reported mechanisms for other bacterial strains are involved in the action of LP14.[viii]
Comment
The probiotic LG2055 showed lowering effects on abdominal adiposity, body weight and other measures, suggesting its beneficial influence on metabolic disorders so state the researchers. Albeit that the improvements are modest (see the table above) the variations may simply reflect the efficiency of their microbial mix and that apart from the proposed bacterially orchestrated direct mechanisms, there may also be a competitive inhibition of the B.Thetaiotaomicron through the oral ingestion of lactic acid bacteria.
Does this mean that all lactic acid bacteria will have this effect? – unlikely, strain specificity is becoming all too clear, but is there a future in the body composition management for our modest companions – I suggest very likely and very soon.
How might we approach a patient with stubborn weight issues? Consider that the bacterial composition may benefit from suitable bacterial supplementation and that the ingestion needs to be daily and for a prolonged period, what is yet to be discovered is should the probiotic be discontinued does the efficiency of storage capacity return?
References
[i] Kadooka Y, Sato M, Imaizumi K, Ogawa A, Ikuyama K, Akai Y, Okano M, Kagoshima M, & Tsuchida T (2010). Regulation of abdominal adiposity by probiotics (Lactobacillus gasseri SBT2055) in adults with obese tendencies in a randomized controlled trial. European journal of clinical nutrition, 64 (6), 636-43 PMID: 20216555
[ii] Bäckhed F, Ding H, Wang T, Hooper LV, Koh GY, Nagy A, Semenkovich CF, & Gordon JI (2004). The gut microbiota as an environmental factor that regulates fat storage. Proceedings of the National Academy of Sciences of the United States of America, 101 (44), 15718-23 PMID: 15505215
[iii] McNeil NI.The contribution of the large intestine to energy supplies in man. Am J Clin Nutr. 1984 Feb;39(2):338-42. View Full Paper
[iv] Flegal KM, Troiano RP. Changes in the distribution of body mass index of adults and children in the US population. Int J Obes Relat Metab Disord. 2000 Jul;24(7):807-18. View Abstract
[v] Ley RE, Turnbaugh PJ, Klein S, Gordon JI. Microbial ecology: human gut microbes associated with obesity. Nature. 2006 Dec 21;444(7122):1022-3. View Abstract
[vi] Kawai Y, Saito T, Uemura J & Itoh T (1997) Rapid detection method for bacteriocin and distribution of bacteriocin-producing strains in Lactobacillus acidophilus group lactic acid bacteria isolated from human feces. Biosci Biotechnol Biochem 61, 179–182. View Full Paper
[vii] Sato M, Uzu K, Yoshida T, Hamad E, Kawakami H, Matsuyama H, Abd El-Gawad I & Imaizumi K (2008) Effects of milk fermented by Lactobacillus gasseri SBT2055 on adipocyte size in rats. Br J Nutr 99, 1013–1017. View Abstract
[viii] Naoki Takemura, Takuma Okubo, & Kei Sonoyama (2010). Lactobacillus plantarum strain No. 14 reduces adipocyte size in mice fed high-fat diet Experimental Biology and Medicine